Advertisement
Grab your lab coat. Let's get started
Welcome!
Welcome!
Create an account below to get 6 C&EN articles per month, receive newsletters and more - all free.
It seems this is your first time logging in online. Please enter the following information to continue.
As an ACS member you automatically get access to this site. All we need is few more details to create your reading experience.
Not you? Sign in with a different account.
Not you? Sign in with a different account.
ERROR 1
ERROR 1
ERROR 2
ERROR 2
ERROR 2
ERROR 2
ERROR 2
Password and Confirm password must match.
If you have an ACS member number, please enter it here so we can link this account to your membership. (optional)
ERROR 2
ACS values your privacy. By submitting your information, you are gaining access to C&EN and subscribing to our weekly newsletter. We use the information you provide to make your reading experience better, and we will never sell your data to third party members.
Pharmaceuticals
Closely Linked
CASE STUDY #1: A unique deal with Siegfried helps Arena Pharmaceuticals advance its first product
by Ann M. Thayer
March 10, 2008
| A version of this story appeared in
Volume 86, Issue 10

NO MILESTONE is more critical for an emerging pharmaceutical company than launching its first product. And although the drug developer may do the heavy lifting throughout R&D and clinical trials, it often turns to a custom manufacturer to make the product at commercial scale. Whether the developer-manufacturer relationship is simply contractual or a close partnership depends on the companies and their shared history.
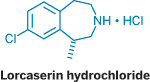
San Diego-based Arena Pharmaceuticals is moving toward launching its initial product. Lorcaserin, a small-molecule antiobesity drug, is in Phase III trials scheduled to end next year. "Things are looking promising," Chief Executive Officer Jack Lief says. Following closely behind lorcaserin in Arena's pipeline are four other development programs, including one each with Ortho-McNeil Pharmaceutical and Merck & Co.
"We are fortunate to have a technology that allows us to discover many promising compounds that specifically target G protein-coupled receptors," Lief says. Arena's GPCR-based discovery technology was developed by its chief scientific officer, Dominic P. Behan, who founded Arena with Lief in 1997. Since then, Lief has been advancing Arena's business strategy.
Arena had considered outsourcing much of its drug manufacturing, but fortuitous timing led to a mutually beneficial deal with one of its active pharmaceutical ingredient (API) suppliers. Now, unlike many start-ups for which production and marketing may be an afterthought or left to a larger pharmaceutical partner, "we have plans to manufacture, and at some point I would also like to see Arena develop its own commercialization capabilities," Lief remarks. "As a company grows, its needs change and our thinking has been to expand our capabilities and take on more and more responsibilities that pharmaceutical companies typically assume."
In late 2007, Arena purchased from Siegfried a facility in Zofingen, Switzerland, that makes drug tablets and other finished dosage forms. The price was about $28 million in cash and 1.5 million shares of Arena stock, worth about $12 million at the time. The Swiss custom manufacturer will become a long-term supplier of bulk lorcaserin and is also developing two other Arena APIs. In turn, Arena will manufacture several finished products for Siegfried for at least three years.
"The initial requirement for an arrangement of this nature is that there be a basis of trust," Siegfried CEO Douglas C. Günthardt says. "It's not just a transaction where everybody goes off their own way but really a long-term partnership." The two companies have worked together for about five years with Siegfried in the role of supplier; now Siegfried is also a customer, and Arena is the most important third-party supplier of its generics division, Günthardt adds.
For Siegfried, the transition is eased by the fact that 70 of its employees will move to Arena and continue to run the finished products facility. Siegfried also will supply quality assurance and other services. The facility has been inspected and approved by both the U.S. Food & Drug Administration and Swiss regulators.
DEALS LIKE THIS happen, Günthardt says, "because senior management is in contact and you realize how the other company is evolving and what they are trying to accomplish." The trigger, he adds, can be when companies are nearing the launch of a new product. Arena was at the point of deciding how to handle its manufacturing requirements, which could be substantial for an antiobesity drug. Siegfried, meanwhile, had recently opened a facility in Malta and could move products there to free up capacity in Zofingen.
Now that it's a manufacturer, Arena is faced with the unenviable challenge of forecasting its production-capacity needs, a volume that could vary widely depending on when and whether lorcaserin will be approved and how well it does. The deal includes a 15-year supply agreement for the API—Arena will have other suppliers as well—but the company is in charge of its own tablet-stage production. The Swiss government gave Arena certain tax benefits for its purchase of the facility.
Balancing its ownership risk, Arena gets a turnkey facility with trained staff and an existing slate of products. "The cost of the agreement with Siegfried is about comparable to what outsourcing would cost us between now and the time that we would begin selling our product," Lief explains. "It also makes so much sense for us because we have multiple drugs in development and eventually we'll need them to be manufactured."
Lief anticipates that having production capabilities may facilitate plans to commercialize lorcaserin with a partner because there will be no need to transfer and validate manufacturing processes. "We'll completely avoid that step by supplying our partner with finished product, which also makes for a much more enhanced regulatory approval process," he adds.
Thanks to reduced manufacturing costs, Arena estimates that the facility's purchase price could be recouped within about a year of launching lorcaserin. It is paying Siegfried about two-thirds of the cash upfront and the rest in installments in the third, fourth, and fifth years after closing. Siegfried, meanwhile, cannot sell its Arena shares for three years.
The CEOs structured the deal in a way that connects the interests of their companies. "We care longer term that Arena is successful, and we are aligned with looking out for them," Günthardt says. "Obviously for us it would be very nice if they do have a drug that gets to market because the upside for the shares could be significant.
"And if their lead compound fails, we also suffer because their share price will take a hit," he acknowledges. "That's the nature of the industry, but they also have other products in the pipeline that might come along within two to three years." He points out that the companies complement each other in their expertise and capabilities and that Arena has shifted from an R&D-focused firm into an integrated operation overnight.
This is not the first time that Siegfried has tailored a deal for a pharmaceutical partner. In 2006, it set up a long-term agreement with Celgene, a New Jersey-based company that it had known for about nine years. The deal revolved around Celgene's second drug, the multiple myeloma treatment Revlimid, which was gaining in sales through approvals in more markets and additional uses.
OVER TIME, Celgene will pay Siegfried about $46 million for production capacity in Zofingen that equals roughly 5% of the Swiss firm's total capacity. Because Revlimid is an analog of the human teratogen thalidomide, the U.S. company wanted to maintain control of drug production by conducting the final steps itself. Siegfried provides intermediates, testing, and support services.
As the companies worked together over the past decade, Celgene launched two major drugs and its product sales grew to $1.3 billion in 2007. Günthardt views Arena, like Celgene, as another promising company on which Siegfried is willing to place a bet.
"There are not many small companies that have the vision or ambition of wanting to be an independent pharmaceutical company," Günthardt remarks about its partners. "But Celgene's management pulled it off, and now Jack Lief has plans that are being executed systematically with that in mind."
Not only was Siegfried's management and board of directors impressed enough to want to partner with Arena, but Günthardt believes his former employees have been impressed as well. A few weeks ago, Lief came to visit them at the finished-dosage facility. "Arena has always said it cares about its people, but when the CEO gets on an airplane and comes out here just to meet them and then go back, they were delighted," he explains. "They may have been a bit cynical about what Americans might say and do, but I think they felt that this guy is basically walking the talk.
"It's another reason we feel good that we have done this deal," Günthardt adds. "We care about the people we have here because they have been good employees and we want to make sure they feel comfortable as well. So we are very pleased with how it has worked out so far."




Join the conversation
Contact the reporter
Submit a Letter to the Editor for publication
Engage with us on Twitter