Advertisement
Grab your lab coat. Let's get started
Welcome!
Welcome!
Create an account below to get 6 C&EN articles per month, receive newsletters and more - all free.
It seems this is your first time logging in online. Please enter the following information to continue.
As an ACS member you automatically get access to this site. All we need is few more details to create your reading experience.
Not you? Sign in with a different account.
Not you? Sign in with a different account.
ERROR 1
ERROR 1
ERROR 2
ERROR 2
ERROR 2
ERROR 2
ERROR 2
Password and Confirm password must match.
If you have an ACS member number, please enter it here so we can link this account to your membership. (optional)
ERROR 2
ACS values your privacy. By submitting your information, you are gaining access to C&EN and subscribing to our weekly newsletter. We use the information you provide to make your reading experience better, and we will never sell your data to third party members.
Physical Chemistry
New N2 Radical Isolated
New complexes protect normally highly unstable anion
by Elizabeth Wilson with reporting by Jyllian Kemsley
July 27, 2009
| A version of this story appeared in
Volume 87, Issue 30

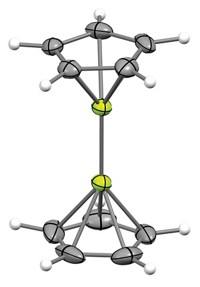
Within the protective framework of exotic metal complexes, scientists have now isolated the reactive, triply reduced N2 3– radical.
In addition to broadening the field of dinitrogen chemistry, the work adds to efforts to develop reactions that could be used in fertilizer-making processes—methods that are milder than the ubiquitous Haber-Bosch method, which reduces N2 under high temperatures and pressures to form ammonia.
The work culminates years of effort from the lab of William J. Evans, chemistry professor at the University of California, Irvine, and colleagues there and at UCLA. In their complexes, the N2 3– anion is bonded to a complex containing potassium as well as a pair of complexes containing either dyspropsium or yttrium (J. Am. Chem. Soc., DOI: 10.1021/ja9036753).
“It is always exciting when new types of N2 compounds are discovered, and the identification of compounds with N2 3– radicals may open unique pathways for dinitrogen functionalization,” notes Paul J. Chirik, a chemistry professor at Cornell University. The yttrium compounds are “particularly striking additions to the library of known N2 compounds,” he adds.
Michael D. Fryzuk, a chemistry professor at the University of British Columbia, Vancouver, calls the work “remarkable chemistry. It is rare that such a completely new form of a small molecule, N2 3–, is uncovered and completely characterized.”
Although dinitrogen complexes obtained by 1-, 2-, and 4-electron reductions have been studied, the N2 3– anion’s radical nature has made it a slippery target. Evans tells C&EN that the unusual metals in the complexes—dysprosium and yttrium, which tend to form ionic bonds, which don’t share their electrons and thus don’t interact very much—made it possible to isolate the ion. In metals that tend to form covalent bonds, the charge density is smeared between two bound atoms, and “the unpaired electron on this ion might communicate better with the surroundings and find another electron with which to pair,” he explains.
Through an exhaustive analysis that included electron spin resonance spectroscopy and Raman spectroscopy, as well as computational validation with density functional theory, Evans says they’ve discovered that the anion’s unpaired electron appears to be in an orbital that is isolated from and protected by the metal-ligand framework.
“This may mimic how nature protects very reactive intermediates,” he says.
N2 3– is electronically analogous to the radical superoxide, O2–, an ion that plays important roles in immune system biochemistry, as well as acting as a source of oxygen on spacecraft and submarines. The electronic resemblance raises a quandary in naming the new ion. The N23– anion isn’t listed in the 2005 IUPAC Nomenclature of Inorganic Chemistry, “presumably because no one thought this species could exist,” Evans says. The group is anticipating how IUPAC will weigh in: Calling it a dinitride? Or, in keeping with the superoxide analogy, perhaps supernitride?


Join the conversation
Contact the reporter
Submit a Letter to the Editor for publication
Engage with us on Twitter