Advertisement
Grab your lab coat. Let's get started
Welcome!
Welcome!
Create an account below to get 6 C&EN articles per month, receive newsletters and more - all free.
It seems this is your first time logging in online. Please enter the following information to continue.
As an ACS member you automatically get access to this site. All we need is few more details to create your reading experience.
Not you? Sign in with a different account.
Not you? Sign in with a different account.
ERROR 1
ERROR 1
ERROR 2
ERROR 2
ERROR 2
ERROR 2
ERROR 2
Password and Confirm password must match.
If you have an ACS member number, please enter it here so we can link this account to your membership. (optional)
ERROR 2
ACS values your privacy. By submitting your information, you are gaining access to C&EN and subscribing to our weekly newsletter. We use the information you provide to make your reading experience better, and we will never sell your data to third party members.
Physical Chemistry
Fullerenes Found In Space
Astrochemistry: Astronomers detect C60 and C70 in dying star
by Elizabeth K. Wilson
July 26, 2010
| A version of this story appeared in
Volume 88, Issue 30
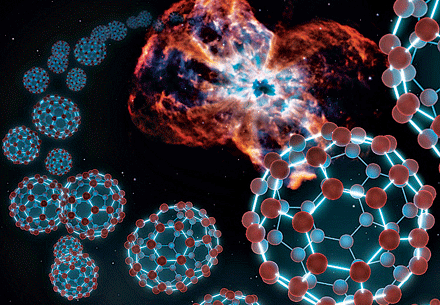
Large amounts of the fullerenes C60 and C70 have been detected in a blast of gas from a dying star—the first unambiguous finding of this form of carbon in space.
The discovery suggests that fullerenes form readily in space, given the right conditions.
Fullerenes, stable and inert, have long been believed to be able to exist in space, particularly near the environment of carbon-rich stars, but evidence has been limited to inconclusive spectra and traces of the molecules in meteorites that fell to Earth.
Astronomer Jan Cami of the University of Western Ontario and his colleagues trained NASA’s Spitzer Space Telescope on a carbon-rich planetary nebula—a star in the late stages of life—and found all of the characteristic infrared emission spectral lines of cold, neutral C60 and C70 (Science, DOI: 10.1126/science.1192035).
Pascale Ehrenfreund, professor of astrobiology at the University of Amsterdam, notes that the detection is “rather unambiguous,” and that these new data are “the best I have seen so far.”
The giant envelopes of gas thrown off by planetary nebulas are rich sources of big molecules, formed by chemical reactions among elements. Many of them are heavy elements such as carbon, which the star has synthesized during its life. These species then go on to populate the interstellar medium.
“The detection of fullerenes and the identification of their formation site are considered one of the priorities in the field of interstellar organic chemistry,” the authors write in their paper.
Previous hunts for fullerenes in planetary nebulas have been difficult because emissions from these stars are often dominated by the strong, complex emissions of another carbon-rich class of molecules, polycyclic aromatic hydrocarbons (PAHs). But in the planetary nebula studied by the authors, there were no signs of PAHs, indicating that the dying star’s environment is unusually hydrogen-poor—an ideal condition for forming fullerenes.
David E. Woon, an astrochemist at the University of Illinois, Urbana-Champaign, points out that the new, recently operational, aircraft-based infrared telescope SOFIA, from NASA and the German Aerospace Center, will be able to add to the study of C60, C70, and other IR-active molecules.





Join the conversation
Contact the reporter
Submit a Letter to the Editor for publication
Engage with us on Twitter