Advertisement
Grab your lab coat. Let's get started
Welcome!
Welcome!
Create an account below to get 6 C&EN articles per month, receive newsletters and more - all free.
It seems this is your first time logging in online. Please enter the following information to continue.
As an ACS member you automatically get access to this site. All we need is few more details to create your reading experience.
Not you? Sign in with a different account.
Not you? Sign in with a different account.
ERROR 1
ERROR 1
ERROR 2
ERROR 2
ERROR 2
ERROR 2
ERROR 2
Password and Confirm password must match.
If you have an ACS member number, please enter it here so we can link this account to your membership. (optional)
ERROR 2
ACS values your privacy. By submitting your information, you are gaining access to C&EN and subscribing to our weekly newsletter. We use the information you provide to make your reading experience better, and we will never sell your data to third party members.
Synthesis
Coupling Hydrazine
Organic Synthesis: Palladium-catalyzed reaction offers new route to heterocycles
by Bethany Halford
October 18, 2010
| A version of this story appeared in
Volume 88, Issue 42

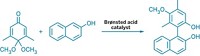
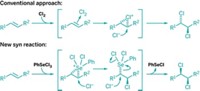
For the first time, chemists have managed to use palladium cross-coupling chemistry to wed aryl groups with hydrazine (Angew. Chem. Int. Ed., DOI: 10.1002/anie.201003764). The resulting aryl hydrazines are useful intermediates en route to nitrogen-containing heterocycles, such as indoles and pyrazoles.
The coupling reaction, developed by chemistry professor Mark Stradiotto and graduate student Rylan J. Lundgren of Dalhousie University, in Nova Scotia, proceeds in moderate to good yields and short reaction times with mild heating. Because hydrazine is a powerful reductant—and is therefore likely to react with both the Pd catalyst and the aryl chloride or aryl tosylate substrate—the transformation relies on electron-rich ligands that circumvent these unwanted side reactions, such as the morpholine-based compound Mor-DalPhos.
“We are still in the process of understanding what makes the Mor-DalPhos ligand such a good ligand for hydrazine cross-coupling,” Stradiotto tells C&EN. “It is a sterically demanding, bidentate ligand, which may help the selectivity of the reaction. The bidentate ligand framework may also help in maintaining a monomeric catalyst species, preventing catalyst decomposition steps, as well as in promoting C–N reductive elimination.”
Although some risk of explosion is associated with heating hydrazine and palladium, Stradiotto says that using hydrazine hydrate rather than anhydrous hydrazine can mitigate the hazard. “We didn’t encounter any difficulties on the scales we performed our reactions [50 mg to 2 g],” he adds, “but it is hard to comment on the potential hazards of the reaction on larger scales.”
“It is certainly a surprise that one can conduct the direct coupling of hydrazine, given how commonly this reagent acts as a reducing agent for the synthesis of Pd(0) complexes,” comments University of Illinois, Urbana-Champaign, chemistry professor John F. Hartwig. “I would have thought one would always have to tame this reagent by conducting the synthesis of N-aryl hydrazines indirectly through hydrazone derivatives.”
John P. Wolfe, a chemistry professor at the University of Michigan, adds, “This work further illustrates that heteroatom nucleophilicity plays a key role in selective Pd-catalyzed carbon-heteroatom bond-forming processes and that relatively small perturbations of ligand structure can have a large impact on catalyst reactivity and selectivity.”



Join the conversation
Contact the reporter
Submit a Letter to the Editor for publication
Engage with us on Twitter