Advertisement
Grab your lab coat. Let's get started
Welcome!
Welcome!
Create an account below to get 6 C&EN articles per month, receive newsletters and more - all free.
It seems this is your first time logging in online. Please enter the following information to continue.
As an ACS member you automatically get access to this site. All we need is few more details to create your reading experience.
Not you? Sign in with a different account.
Not you? Sign in with a different account.
ERROR 1
ERROR 1
ERROR 2
ERROR 2
ERROR 2
ERROR 2
ERROR 2
Password and Confirm password must match.
If you have an ACS member number, please enter it here so we can link this account to your membership. (optional)
ERROR 2
ACS values your privacy. By submitting your information, you are gaining access to C&EN and subscribing to our weekly newsletter. We use the information you provide to make your reading experience better, and we will never sell your data to third party members.
Biological Chemistry
Molecular Machine’s Workings Visualized
Study provides real-time glimpse of assembly of single spliceosomes
by Stu Borman
March 21, 2011
| A version of this story appeared in
Volume 89, Issue 12

A new imaging technique can be used to understand the workings of complex biomolecular processes at a more detailed level than previously possible.
The technique marks biomolecules with organic dyes based on new chemical tagging technology and uses lasers and fluorescence microscopy to analyze the molecules’ activities. Its developers have used it to provide a glimpse of the way a huge biomolecular machine, the spliceosome, works in real time, at a single-molecule level, and in a near-native environment.
The technique has now given scientists a deeper understanding of the assembly of component parts of the spliceosome—which controls the diversity of proteins produced in cells and plays a role in cancer and other diseases. The approach could be used to determine currently unknown details of other complex biomolecular processes, such as DNA replication and protein translation. The spliceosome assembly findings could also help lead to new therapies for conditions caused by spliceosome malfunctions.
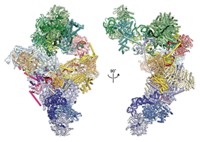
The spliceosome is a huge protein and RNA complex that is responsible for editing pre-messenger RNA (pre-mRNA) to mature mRNA. Pre-mRNA is nascent mRNA that has been newly transcribed from genomic DNA. The spliceosome edits it by snipping out intervening sequences that are not needed in translated proteins and linking together the remaining parts of pre-mRNA. The resulting edited mRNAs are then translated into working proteins.
One aspect of the editing process, called alternative splicing, makes it possible for each gene in the body to code for numerous proteins and thus allows an organism’s proteins to be much more varied than the number of its genes would seem to allow. In humans, more than 90% of genetically encoded pre-mRNAs are alternatively spliced into mature mRNAs.
The new technique makes it possible to learn more about how the spliceosome works at a detailed molecular level. The technique was developed and demonstrated in a collaboration among Melissa J. Moore of Howard Hughes Medical Institute and the University of Massachusetts Medical School, Worcester; Jeff Gelles of Brandeis University; Virginia W. Cornish of Columbia University; and coworkers (Science, DOI: 10.1126/science.1198830).
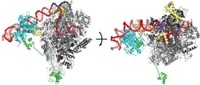
In the five-year study, postdoctoral researcher Aaron A. Hoskins labeled individual components of the yeast spliceosome with organic fluorophores using a new chemical tagging technology developed by Cornish’s group. The multiwavelength fluorescent tags were designed to provide 10 times higher photon output than fluorescent proteins, enabling simultaneous detection of different components at a single-molecule level. Hoskins then used a version of colocalization single-molecule spectroscopy developed by Gelles’ group to monitor the components as they assembled together in lysed yeast cell extracts to form working spliceosomes. Moore is the collaborative team’s spliceosome expert.
The approach made it possible to visualize directly, in real time, the ordered process in which the small nuclear ribonucleoproteins U1 and U2, the tri-small nuclear ribonucleoprotein U4/U6.U5, and the multiprotein Prp19 complex join together with pre-mRNA to form spliceosomes.
The broad outline of this assembly process was already known from previous studies. But the new study shows for the first time that the addition of each spliceosome component to the overall complex is reversible and that assembly can therefore proceed in both forward and backward directions. In addition, it reveals that assembly is not preordained but rather becomes increasingly more committed to completion as additional components are added.
“The single-molecule analysis of spliceosome assembly described in this paper is a powerful method that will certainly lead to new, high-impact insights,” says Magda Konarska, a spliceosome specialist at Rockefeller University. “I am really looking forward to future developments—there is a potential here for verifying some existing ideas and formulating new ways of thinking about spliceosome assembly.”
“Some of the study’s findings are very much in line with what was known before from much more cumbersome genetic and ensemble in vitro experiments,” says Nils G. Walter of the University of Michigan, Ann Arbor, who specializes in single-molecule analysis of RNA and the spliceosome. “But it wasn’t clear before that in the sequential addition of components to the spliceosomal complex there is reversibility at each step, which allows for proofreading and corrections along the way. This lays the foundation for models showing how splicing could be dynamically regulated, giving different mRNA products and translated proteins depending on the functional state of a particular cell.”


Join the conversation
Contact the reporter
Submit a Letter to the Editor for publication
Engage with us on Twitter