Advertisement
Grab your lab coat. Let's get started
Welcome!
Welcome!
Create an account below to get 6 C&EN articles per month, receive newsletters and more - all free.
It seems this is your first time logging in online. Please enter the following information to continue.
As an ACS member you automatically get access to this site. All we need is few more details to create your reading experience.
Not you? Sign in with a different account.
Not you? Sign in with a different account.
ERROR 1
ERROR 1
ERROR 2
ERROR 2
ERROR 2
ERROR 2
ERROR 2
Password and Confirm password must match.
If you have an ACS member number, please enter it here so we can link this account to your membership. (optional)
ERROR 2
ACS values your privacy. By submitting your information, you are gaining access to C&EN and subscribing to our weekly newsletter. We use the information you provide to make your reading experience better, and we will never sell your data to third party members.
Physical Chemistry
Reacting By Roaming
Studies strengthen role for alternative to transition-state pathways
by Jyllian Kemsley
March 19, 2012
| A version of this story appeared in
Volume 90, Issue 12
Transition-state theory is one of the bedrocks of chemistry. Even introductory students might see a plot of potential energy versus reaction coordinate, with reactants on the left, products on the right, and a high energy barrier in between. But researchers increasingly see dissociation or decomposition reactions bypassing their conventional transition-state routes in favor of what’s called a “roaming” mechanism.
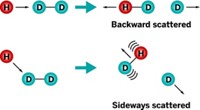
In roaming, an atom or group pulls away from the rest of a molecule, as if to dissociate the molecule into two radical species. But instead of fully separating, the mobile piece moves out just enough to give itself some freedom. It can then move about in the vicinity of the remaining atoms.
In formaldehyde photodissociation, for example, a C–H bond may stretch out to about 3 to 4 Å—more than three times its normal length—allowing the hydrogen to start roaming around. If it then encounters the other hydrogen and abstracts it, the reaction will yield H2 and CO. In acetaldehyde, the methyl group pulls away to roam, producing CH4 and CO.

The effects can be mapped on the potential energy surfaces of the reactions. Even if the narrow path through a specific conventional transition state might be the lowest energy route, such paths are also constricted. The reaction pays a high price in entropy so the tight pathway is disfavored. Roaming paths, in contrast, are wider, looser, and offer molecules more flexibility.
The end products of roaming are the same as those formed through conventional transition-state pathways. Scientists can determine which route a reaction follows because the different paths yield molecules with different rotational or vibrational energies.
The dissociations of formaldehyde and acetaldehyde were the first two reactions found to occur through roaming mechanisms, in 2004 and 2006. Since then, researchers have also documented roaming in the thermal decomposition of alkanes and dimethyl ether, as well as dissociation of MgH2. In all of these cases, the molecules also follow conventional transition-state pathways, and both mechanisms contribute to the product pool.
Recent work, however, shows that some reactions proceed exclusively through roaming. In one study, Wayne State University chemistry professor Arthur G. Suits and colleagues looked at the photodissociation of nitrobenzene (C6H5NO2). They found that roaming of the NO2 group isomerizes the molecule to phenyl nitrite (C6H5ONO) before the molecule breaks up into C6H5O• and NO• (Nat. Chem., DOI: 10.1038/nchem.1194 ).
In another study, Texas A&M University chemistry professor Simon W. North and coworkers looked at the nitrate radical (NO3•), a key atmospheric oxidant that photodissociates to either NO2• and O• or NO• and O2. Although scientists have studied NO3• chemistry since 1880, “it’s really been a bit of a mystery how you get NO• and O2 from NO3•,” North says. He and colleagues found that the reaction happens only through roaming. They also found that NOreacts from both its ground state and an excited state, something not previously observed (Science, DOI: 10.1126/science.1216911).
The result “broadens our perspective on how reactions can happen,” North says. “We can begin to ask questions about when would such things happen in other systems.”
A better understanding of roaming may have a number of important applications. For example, the ignition temperature for combustion is determined by the threshold for making radicals. Because roaming turns potential radicals into molecular products, roaming mechanisms have now been incorporated into combustion models, says Joel M. Bowman, a chemistry professor at Emory University. Bowman studied formaldehyde roaming with Suits and acetaldehyde roaming with University of Sydney chemistry professor Scott H. Kable and Sandia National Laboratories researcher David L. Osborn.
Roaming also brings up some intriguing questions, such as whether a roaming atom or group can still be considered bound to its originating molecule, says Texas Tech University chemistry professor William L. Hase. Hase adds that the long-range, weak interactions of the molecular fragments will also be a new and interesting area to explore. Hase and colleagues found a variation of roaming in the nucleophilic substitution reaction of Cl– with CH3I. In the roaming component of that reaction, the Cl– first collides with the side of the CH3 group, causing it to rotate around the I atom. Then the Cl– comes in again to the back of the CH3 group to displace I–.
“We are indeed coming to the conclusion that these roaming mechanisms are quite common,” says Lawrence B. Harding, a research fellow at Argonne National Laboratory who has done several computational studies on roaming and is now investigating alkyl halides. He and others emphasize that the reason roaming mechanisms hadn’t been identified earlier is simply because people hadn’t looked in the parts of potential energy surfaces where the roaming pathways lie.
Adds Suits, “If we had just looked at and thought about this 50 years ago with an open mind, we could have anticipated roaming easily.”



Join the conversation
Contact the reporter
Submit a Letter to the Editor for publication
Engage with us on Twitter