Advertisement
Grab your lab coat. Let's get started
Welcome!
Welcome!
Create an account below to get 6 C&EN articles per month, receive newsletters and more - all free.
It seems this is your first time logging in online. Please enter the following information to continue.
As an ACS member you automatically get access to this site. All we need is few more details to create your reading experience.
Not you? Sign in with a different account.
Not you? Sign in with a different account.
ERROR 1
ERROR 1
ERROR 2
ERROR 2
ERROR 2
ERROR 2
ERROR 2
Password and Confirm password must match.
If you have an ACS member number, please enter it here so we can link this account to your membership. (optional)
ERROR 2
ACS values your privacy. By submitting your information, you are gaining access to C&EN and subscribing to our weekly newsletter. We use the information you provide to make your reading experience better, and we will never sell your data to third party members.
Environment
Water-Catalyzed Nanofabrication Strikes Gold
Hot, levitating waterdrops catalyze formation of gold nanoparticles, nanocoatings
by
November 4, 2013
| A version of this story appeared in
Volume 91, Issue 44
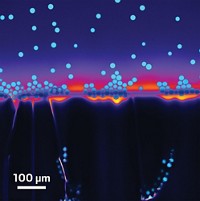
Hot off the frying pan may be a simple, green way to make nanomaterials. Water droplets levitating on a hot plate can catalyze formation of small amounts of nanomaterials, including gold nanoparticles and nanocoatings, without the need for other reagents, report Mady Elbahri and Ramzy Abdelaziz of Germany’s University of Kiel and colleagues (Nat. Commun. 2013, DOI: 10.1038/ncomms3400). The method exploits the Leidenfrost effect, in which water droplets on hot surfaces, such as frying pans, levitate as molecules near the surface quickly vaporize. The study showed that a drop of aqueous tetrachloroauric acid solution on a 270 °C hot plate levitated and morphed into a suspension of gold nanoparticles. Such particles are used in biomedical and other applications, but their synthesis often requires toxic reagents. The researchers also used the method to create zinc oxide and copper oxide nanoparticles and coat them on a three-dimensional object—an electron microscope specimen grid. These materials did not form at sub-Leidenfrost temperatures, highlighting the effect’s crucial role. The researchers suggest that when the hot water levitates, it becomes negatively charged, creating favorable reaction conditions. It’s not yet clear whether the method can be scaled up, they say.



Join the conversation
Contact the reporter
Submit a Letter to the Editor for publication
Engage with us on Twitter