Advertisement
Grab your lab coat. Let's get started
Welcome!
Welcome!
Create an account below to get 6 C&EN articles per month, receive newsletters and more - all free.
It seems this is your first time logging in online. Please enter the following information to continue.
As an ACS member you automatically get access to this site. All we need is few more details to create your reading experience.
Not you? Sign in with a different account.
Not you? Sign in with a different account.
ERROR 1
ERROR 1
ERROR 2
ERROR 2
ERROR 2
ERROR 2
ERROR 2
Password and Confirm password must match.
If you have an ACS member number, please enter it here so we can link this account to your membership. (optional)
ERROR 2
ACS values your privacy. By submitting your information, you are gaining access to C&EN and subscribing to our weekly newsletter. We use the information you provide to make your reading experience better, and we will never sell your data to third party members.
Materials
Nanocomposite Material Help Capacitors Pack A High Energy Punch
Electronics: Polymer-nanowire combination leads to electrical components that store large amounts of energy in small volumes
by Katherine Bourzac
March 15, 2013

In electrical circuits that drive lasers, run cardiac implants, or convert between alternating and direct current, components called capacitors produce large, fast pulses of energy. A new composite material containing a fluorinated polymer and ceramic nanowires allows capacitors to store more energy in a smaller volume than commercial devices can (Nano Lett., DOI: 10.1021/nl3037273). The materials could help engineers to reduce the size of these components and cut production costs.
Capacitors store static charge in a layer of insulating material sandwiched between conductive plates. A capacitor’s size dictates how much energy it can store. So components that provide large amounts of energy tend to be large and heavy, says Henry A. Sodano, a materials scientist at the University of Florida.
Sodano and other researchers want to improve capacitors’ insulating materials to pack more energy in smaller volumes. Capacitors with high energy densities could make electrical systems lighter in hybrid vehicles and lower production costs of power conversion equipment that connect solar and wind farms to power transmission grids.
To shrink high energy capacitors, researchers are trying to increase both the amount of charge a capacitor can store in a given volume and the voltage at which it can operate. Unfortunately, doing both is tricky.
Some commercial capacitor materials, such as polypropylene, have a high breakdown strength, meaning they can function at high voltages. But these materials store relatively small amounts of charge per volume, so capacitors made with them must be large to carry more energy. Attempts to improve the charge storage capacity of these materials have lowered their breakdown strengths, leading to little net benefit in energy storage, Sodano says.
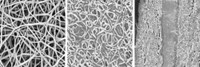
He and his team developed a new composite material that improved charge storage but didn’t compromise much on breakdown strength. The material consists of a high breakdown strength polymer called polyvinylidene fluoride and nanowires of barium strontium titanium oxide. The oxide is a ceramic that’s prized for its charge-storing properties and is often found in capacitors. The researchers decided to use nanowires of the ceramic because theoretical calculations suggested that the shape would optimize charge storage. They produced thin films of the nanocomposite material by casting a solution of the ceramic nanowires and polymer onto a glass plate and letting it dry.
Capacitors made from the material had breakdown strengths comparable to commercial polymer capacitors: 450 MV/m and 640 MV/m, respectively. The nanocomposite capacitors stored more energy per volume than the commercial components, 14.86 J/cm3 compared to 1.2 J/cm3. “It’s like a power plant stored in a cubic centimeter,” Sodano says.
Michael Lanagan, a materials scientist at Pennsylvania State University, calls the composite material an interesting way to reduce the size of capacitors. However, Lanagan notes that other experimental materials, including glasses tested in his lab, have exhibited even greater breakdown strength and energy storage. The challenge for groups working on new capacitor materials, he says, will be to not only improve the electrical properties, but also make them compatible with large-scale manufacturing.




Join the conversation
Contact the reporter
Submit a Letter to the Editor for publication
Engage with us on Twitter