Advertisement
Grab your lab coat. Let's get started
Welcome!
Welcome!
Create an account below to get 6 C&EN articles per month, receive newsletters and more - all free.
It seems this is your first time logging in online. Please enter the following information to continue.
As an ACS member you automatically get access to this site. All we need is few more details to create your reading experience.
Not you? Sign in with a different account.
Not you? Sign in with a different account.
ERROR 1
ERROR 1
ERROR 2
ERROR 2
ERROR 2
ERROR 2
ERROR 2
Password and Confirm password must match.
If you have an ACS member number, please enter it here so we can link this account to your membership. (optional)
ERROR 2
ACS values your privacy. By submitting your information, you are gaining access to C&EN and subscribing to our weekly newsletter. We use the information you provide to make your reading experience better, and we will never sell your data to third party members.
Environment
Milk Proteins Protect Fabrics From Fire
Flame Retardants: Casein, a protein left over from cheese production, could be an ecofriendly and nontoxic alternative to current phosphate-based retardants
by Alexander Hellemans
March 11, 2014

Many compounds currently used as flame retardants in upholstered furniture and plastics have come under scrutiny for their potentially harmful effects on the environment and human health. Since manufacturers often rely on flame retardants to meet fire-safety standards for certain products, many have looked for nontoxic compounds as alternatives. Now, researchers in Italy have demonstrated that caseins—proteins found in milk that are a by-product of cheese production—may fit the bill (Ind. Eng. Chem. Res. 2014, DOI: 10.1021/ie404089t).
Some types of flame retardants, such as organophosphate esters, get their fire-blocking properties from their high phosphorus content. When they burn, a polymer layer of phosphoric acid forms and creates a char that blocks heat transfer to unburned areas of the material, slowing the spread of the fire.
Jenny Alongi of the Polytechnic University of Turin and her colleagues decided to investigate a family of proteins called caseins as alternative flame retardants because they contain a large number of phosphate groups. Caseins are found in the whey that’s a by-product of cheese production, so in countries that produce a lot of cheese, such as Italy and France, the proteins are cheap and abundant, Alongi says.
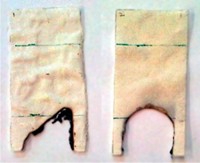
The team coated three materials—cotton, polyester, and a blend of 65% polyester and 35% cotton—with the proteins by soaking the fabrics in distilled water mixed with casein powder. The researchers then submitted the samples to a battery of flammability tests. The results were encouraging: In cotton- and polyester-only fabrics treated with caseins, flames extinguished themselves, leaving 86% of the cotton and 77% of the polyester unburned. The cotton-polyester blend burned completely but took 60% more time to do so than the untreated material.
The flame-retardant properties of caseins also compared well to those of ammonium polyphosphate (APP), a flame retardant used for fire proofing polyolefins and polyurethanes. The caseins effectively form a char layer on the fabric samples. But unlike APP, they don’t produce toxic fumes during combustion.
Before caseins can be used as flame retardants, researchers need to work out many issues, such as preventing the proteins from washing off materials. The team is now testing light-curable resins and molecules such as urea that could bond the casein molecules to the surface of the fabric, Alongi says. Another problem is that materials treated with caseins smell rancid. Alongi and her colleagues are looking for ways to remove the molecules associated with casein that produce the odor.
The caseins performed well compared to current flame retardants, says Jacob de Boer, an analytical chemist at VU University Amsterdam, who has investigated the impact of organophosphorus flame retardants on the environment. He hopes the team pursues the research further.



Join the conversation
Contact the reporter
Submit a Letter to the Editor for publication
Engage with us on Twitter