Advertisement
Grab your lab coat. Let's get started
Welcome!
Welcome!
Create an account below to get 6 C&EN articles per month, receive newsletters and more - all free.
It seems this is your first time logging in online. Please enter the following information to continue.
As an ACS member you automatically get access to this site. All we need is few more details to create your reading experience.
Not you? Sign in with a different account.
Not you? Sign in with a different account.
ERROR 1
ERROR 1
ERROR 2
ERROR 2
ERROR 2
ERROR 2
ERROR 2
Password and Confirm password must match.
If you have an ACS member number, please enter it here so we can link this account to your membership. (optional)
ERROR 2
ACS values your privacy. By submitting your information, you are gaining access to C&EN and subscribing to our weekly newsletter. We use the information you provide to make your reading experience better, and we will never sell your data to third party members.
Analytical Chemistry
Microfluidic Chip Enables Fast Extraction Of Scarce Metabolites
Metabolomics: Chip miniaturizes method for enriching low-abundance analytes in a sample, offering fast way to increase limits of detection
by Puneet Kollipara
July 1, 2014
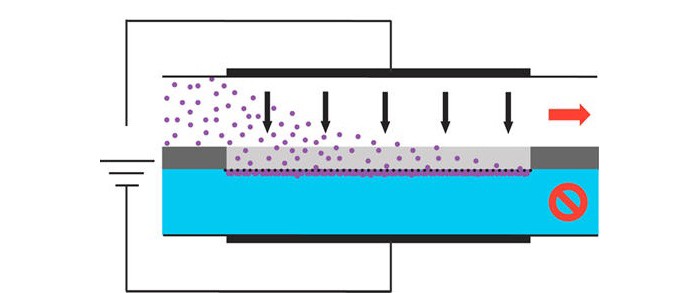
When looking for charged metabolites in urine or other biological samples, researchers often use a technique called electroextraction to enrich the biomarkers’ concentrations, making detection easier. Current electroextraction methods can process only tens of microliters of sample at a time. Although processing larger sample volumes could improve limits of detection for metabolites, doing so currently is time-consuming. Now, researchers have paired electroextraction with a microfluidic chip to process a milliliter or more of a continuously flowing sample in minutes (Anal. Chem. 2014, DOI: 10.1021/ac500707v).
In electroextraction, researchers first dilute the sample into an organic solvent, such as ethyl acetate, that is immiscible with water. Then they add this solvent to a well or capillary tube along with an aqueous solution. They apply an electric field across the liquid sample, forcing the metabolites into the adjacent aqueous solution for collection. This concentrates the analytes into a smaller volume, making their subsequent detection easier. Processing larger volumes of sample means more metabolites land in the small volume of aqueous solution, thus improving the limit of detection. But with larger volumes, analytes have to travel longer distances, making the process more time-consuming.
To cut down on the distance the metabolites have to diffuse through, Petrus W. Lindenburg and his colleagues at Leiden University, in the Netherlands, miniaturized an electroextraction system in a microfluidic chip. Fresh sample flows continuously through the device, replenishing depleted sample to boost the volume that can be processed in one run. Sample flows through the device at a rate of 50 μL per minute, so researchers could extract 1 mL of sample in about 20 minutes.
The team tested the method on one group of biomolecules known as acylcarnitines, a biomarker of health problems such as fatty acid oxidation disorders and diabetes. The team ran an ethyl acetate solution containing urine that they had spiked with acylcarnitines through the system. After the electroextraction, they used liquid chromatography and mass spectrometry to detect the metabolites. The researchers found that the method extracted not only the acylcarnitines they had added but also different ones found in the urine. The detection limit ranged from 0.3 to 2 nM.
The microfluidics chip could enable researchers to almost fully automate the extraction process, further boosting its appeal, says Michael C. Breadmore, an analytical chemist at the University of Tasmania, in Australia. The method might work well with molecules such as amines, alcohols, and peptides, which either are naturally charged or can be given a charge in the lab, he says. But the method may not work with difficult-to-charge molecules, such as sugars.



Join the conversation
Contact the reporter
Submit a Letter to the Editor for publication
Engage with us on Twitter