Advertisement
Grab your lab coat. Let's get started
Welcome!
Welcome!
Create an account below to get 6 C&EN articles per month, receive newsletters and more - all free.
It seems this is your first time logging in online. Please enter the following information to continue.
As an ACS member you automatically get access to this site. All we need is few more details to create your reading experience.
Not you? Sign in with a different account.
Not you? Sign in with a different account.
ERROR 1
ERROR 1
ERROR 2
ERROR 2
ERROR 2
ERROR 2
ERROR 2
Password and Confirm password must match.
If you have an ACS member number, please enter it here so we can link this account to your membership. (optional)
ERROR 2
ACS values your privacy. By submitting your information, you are gaining access to C&EN and subscribing to our weekly newsletter. We use the information you provide to make your reading experience better, and we will never sell your data to third party members.
Environment
Oil Dispersant Compound Persists For Years After Gulf Spill
Environmental Toxicology: Traces of dispersants applied in the Deepwater Horizon spill remain months later in deep-sea coral communities and years later on coastlines
by Deirdre Lockwood
July 9, 2014

Nearly four years after the Deepwater Horizon oil spill in 2010, traces of dispersants, the chemicals applied to break up the oil, remain along the Gulf coastline, a team of scientists report. Dispersant compounds also persisted for at least six months after the spill in deep-sea coral communities in the Gulf (Environ. Sci. Technol. Lett. 2014, DOI: 10.1021/ez500168r).
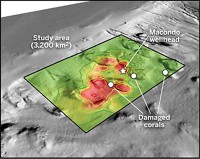
In an effort to keep oil from forming surface slicks during the spill, cleanup crews applied nearly 2 million gallons of dispersant by spraying it on the sea surface and injecting it at depth near the leaking wellhead of BP’s Macondo Well. The dispersant formulations, Corexit 9527 and 9500A, contain surfactants and hydrocarbon solvents that help break up the oil into small droplets to speed up its degradation by microbes.
Based on earlier laboratory studies, researchers expected the dispersant itself to break down quickly in the environment (Environ. Sci. Technol. 2013, DOI: 10.1021/es303881h). But because it is toxic to marine life at certain concentrations, scientists wanted to know whether it fully degraded after the spill response.

So six months after the spill, Helen K. White of Haverford College and her colleagues sampled seafloor sediment around two communities of deep-sea corals within 15 miles of the wellhead. They also collected oil residue from one community, which was covered in a flocculent material containing weathered oil. The coral showed signs of stress, including several dead branches. Then, over a period of two to four years after the spill, the researchers gathered samples of oil-soaked sand, including patties and tar balls, that had washed up along the Gulf coastline at seven sites from Louisiana to Florida.
They analyzed all the samples for the presence of crude oil and a major active ingredient of the dispersant, the anionic surfactant dioctyl sodium sulfosuccinate (DOSS). Using a method developed by their colleagues at Woods Hole Oceanographic Institution, the researchers extracted the samples with a methanol-water solution and analyzed them using liquid chromatography with tandem mass spectrometry.
At all deep-sea and coastal sites but one, they found the signature of oil from the Deepwater Horizon spill. DOSS was present at many of the sites, though at highly variable concentrations. In the deep sea, they found traces of DOSS in the oil residue on the ailing corals at concentrations of 6 to16 ng/g, and at the two sediment sites between 19 and 9,000 ng/g. On the coastline, they found DOSS in nearly all samples of oil-soaked sand at concentrations of 1 to 260 ng/g.
The researchers want to assess the compound’s toxicity to marine organisms at these concentrations in sediments because little is known about their effects in these contexts, White says. The Environmental Protection Agency reports that DOSS may be harmful to marine organisms at concentrations above 40 μg/L in water, but the agency has no such benchmark for levels in sediments and sands.
White plans to study how the oil and dispersant combine in the environment and how this affects the compound’s persistence and bioavailability to marine life.
“We were unaware that these compounds persist in the beach environment,” says Jennifer A. Field, an environmental chemist at Oregon State University, who is also studying the persistence of DOSS after the Gulf spill. She says more work needs to be done to assess the full extent of the dispersant’s distribution and its environmental impact.



Join the conversation
Contact the reporter
Submit a Letter to the Editor for publication
Engage with us on Twitter