Advertisement
Grab your lab coat. Let's get started
Welcome!
Welcome!
Create an account below to get 6 C&EN articles per month, receive newsletters and more - all free.
It seems this is your first time logging in online. Please enter the following information to continue.
As an ACS member you automatically get access to this site. All we need is few more details to create your reading experience.
Not you? Sign in with a different account.
Not you? Sign in with a different account.
ERROR 1
ERROR 1
ERROR 2
ERROR 2
ERROR 2
ERROR 2
ERROR 2
Password and Confirm password must match.
If you have an ACS member number, please enter it here so we can link this account to your membership. (optional)
ERROR 2
ACS values your privacy. By submitting your information, you are gaining access to C&EN and subscribing to our weekly newsletter. We use the information you provide to make your reading experience better, and we will never sell your data to third party members.
Reaction Mechanisms
Easy access to boronate esters and boronic acids
Methodology swaps out carboxylic acids for boron functional groups at late stages of syntheses
by Bethany Halford
April 13, 2017
| A version of this story appeared in
Volume 95, Issue 16

Boron—the beloved oddball of the periodic table—can do some unique chemistry, thanks to its empty p-orbital. This has made boronic acids and boronate esters indispensable components in many compounds, such as in partners for Suzuki cross-coupling reactions, in polymers, and even in a few drugs, such as the cancer therapies bortezomib (Velcade) and ixazomib (Ninlaro). But, while there are a few ways to make boronic acids and boronate esters, none are trivial.
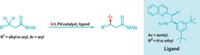
Phil S. Baran’s group at Scripps Research Institute, California, has now come up with a simple and practical method for converting carboxylic acids into a boronate esters and boronic acids (Science 2017, DOI: 10.1126/science.aam7355).
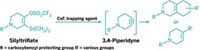
The method works on primary, secondary, and tertiary carboxylic acids, as well as peptidic and even natural product-derived substrates. It also tolerates a broad range of functional groups, so it can be used in the final steps of the synthesis of a densely functionalized molecule. For example, Baran’s group prepared a boronic acid analog of the antibiotic vancomycin, a compound that is packed with stereocenters and functional groups. The reaction conditions modified only vancomycin’s carboxylic acid.
To convert a carboxylic acid to a boron compound, Baran’s team first transforms it into a redox-active N-hydroxyphthalimide ester. This compound then undergoes decarboxylative borylation in the presence of a nickel catalyst, a bipyridine ligand, and bis(pinacolato)diboron to make the boronate ester. These two reactions can be carried out in a single pot. The boronate esters can then be hydrolyzed to the corresponding boronic acids.
The new method “will see immediate application in laboratories throughout academia and the pharmaceutical industry,” comments Stephen L. Buchwald, a synthetic organic chemist at MIT. “The demonstrated substrate scope and the complexity of the applications that are described are spectacular.”
“I particularly like the opportunity for late-stage transformation of more complex molecules,” adds Derek Lowe, a medicinal chemist who writes the popular “In the Pipeline” blog. “Sticking in a boronic acid at that point, though desirable, isn’t what you’d normally consider doing because of the synthetic difficulties.”
In another demonstration of the method’s power, Baran’s group teamed up with scientists at Calibr to make boronic acid analogs of human neutrophil elastase inhibitors. Neutrophil elastase is an important target in lung diseases, such as cystic fibrosis and chronic obstructive pulmonary disease. Calibr had been working with inhibitors that had failed Phase II clinical trials because of potency issues. Baran group’s chemistry added a boronic acid to these molecules and the resulting compounds proved to be far more potent than the parent compounds in tests, including enzyme assays. Calibr is currently studying the in vivo activity of the compounds.
“I always tell people that I barely know what DNA stands for and it’s hard for me to imagine making real contributions to biology or medicine,” Baran says. “But we do know organic chemistry. So this is an example of a very fundamental organic chemistry problem that has immediate ramifications in many areas—materials, medicine, fine chemicals. There are tons of practical applications.”



Join the conversation
Contact the reporter
Submit a Letter to the Editor for publication
Engage with us on Twitter