Advertisement
Grab your lab coat. Let's get started
Welcome!
Welcome!
Create an account below to get 6 C&EN articles per month, receive newsletters and more - all free.
It seems this is your first time logging in online. Please enter the following information to continue.
As an ACS member you automatically get access to this site. All we need is few more details to create your reading experience.
Not you? Sign in with a different account.
Not you? Sign in with a different account.
ERROR 1
ERROR 1
ERROR 2
ERROR 2
ERROR 2
ERROR 2
ERROR 2
Password and Confirm password must match.
If you have an ACS member number, please enter it here so we can link this account to your membership. (optional)
ERROR 2
ACS values your privacy. By submitting your information, you are gaining access to C&EN and subscribing to our weekly newsletter. We use the information you provide to make your reading experience better, and we will never sell your data to third party members.
Analytical Chemistry
Enzyme harnesses light to make alkanes
Newly identified enzyme powers fatty acid decarboxylation with blue light
by Jyllian Kemsley
August 31, 2017
| A version of this story appeared in
Volume 95, Issue 35

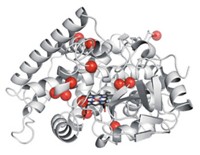
Although many organisms respond to light, very few enzymes inside those organisms use light directly as an energy source for catalysis. Researchers now report a new light-driven enzyme: Christened fatty acid photodecarboxylase (FAP), it uses blue light to drive the removal of carboxyl groups from fatty acids to form alkanes or alkenes (Science 2017, DOI: 10.1126/science.aan6349).

FAP joins a select group of so-called photoenzymes, including DNA-repair enzymes called photolyases, that use light for catalysis on their own rather than functioning as part of a larger complex such as photosystems I or II, which are used by plants and algae for photosynthesis.
The team that identified FAP was led by Fred Beisson, a scientist in the Biosciences & Biotechnologies Institute of the French Alternative Energies & Atomic Energy Commission. Beisson and colleagues knew that some species of microalgae could convert long-chain fatty acids to alkanes or alkenes through a light-dependent mechanism and that the microalgae didn’t possess any genes similar to those that code for hydrocarbon-synthesizing enzymes in other organisms. That puzzle eventually led them to FAP. “Initially, we thought that the synthesis of the enzyme or its mRNA was regulated by light,” Beisson says. The team “didn’t imagine” that the enzyme itself would directly use light for catalysis, he says.
FAP contains flavin adenine dinucleotide (FAD), which commonly serves as a redox cofactor in biological reactions. In the case of FAP, however, FAD absorbs blue light to reach an excited state that abstracts an electron from the carboxylate group of a C12to C18 fatty acid, which then decarboxylates to yield an alkane or alkene.
“This and other recent discoveries suggest a hitherto hidden scope for biotechnologically useful photochemistry by means of cofactor-dependent enzymes,” writes Nigel S. Scrutton, director of the Manchester Institute of Biotechnology, in a commentary accompanying the report.
Beisson now wonders whether other flavoenzymes are unidentified photoenzymes. When scientists measure enzyme activity, he says, turning off the lights to see if the enzyme still works “isn’t something you usually think about.”



Join the conversation
Contact the reporter
Submit a Letter to the Editor for publication
Engage with us on Twitter