Advertisement
Grab your lab coat. Let's get started
Welcome!
Welcome!
Create an account below to get 6 C&EN articles per month, receive newsletters and more - all free.
It seems this is your first time logging in online. Please enter the following information to continue.
As an ACS member you automatically get access to this site. All we need is few more details to create your reading experience.
Not you? Sign in with a different account.
Not you? Sign in with a different account.
ERROR 1
ERROR 1
ERROR 2
ERROR 2
ERROR 2
ERROR 2
ERROR 2
Password and Confirm password must match.
If you have an ACS member number, please enter it here so we can link this account to your membership. (optional)
ERROR 2
ACS values your privacy. By submitting your information, you are gaining access to C&EN and subscribing to our weekly newsletter. We use the information you provide to make your reading experience better, and we will never sell your data to third party members.
Green Chemistry
A more natural approach to catalysts
To make chemical processing more sustainable, chemists turn to animal, vegetable, and mineral sources for catalytic materials
by Stephen K. Ritter
February 20, 2017
| A version of this story appeared in
Volume 95, Issue 8
As a teenager in the 1960s, Charles V. Johnson of Lake Geneva, Wis., was tinkering with his chemistry set when he discovered that earwax could serve as a catalyst for making pigments. Later on, as a zoology undergraduate student at the University of Wisconsin, Madison, Johnson took a daring chance in a chemistry lab: He applied earwax to a boiling chip and substituted it for a palladium catalyst in an organic synthesis experiment. It worked well to make trans-stilbene, although his professor didn’t seem impressed.
“That’s the thing that has bothered me most,” Johnson told C&EN in a 2012 interview. “My instructors didn’t think there was anything to it.” After graduating, Johnson worked as a chemical technician at Sigma-Aldrich until he retired. He occasionally toyed with using earwax as a catalyst over the years to, for example, polymerize a methacrylate-based material he bummed off his dentist.
Johnson often contemplated what the active catalyst might be in earwax, but he wasn’t able to do an analysis to find out. Most likely, it’s an amino acid or protein, he assumed. Amino acids such as proline are well-known organocatalysts. And catalytic proteins, known as enzymes, have been used since the dawn of civilization—though not knowingly until modern times—for food and beverage processing.
Chemists such as Johnson are “natural” experimentalists in every sense. And given that nature provides chemists everything they need to do experiments, it’s only natural that when it comes to looking for ways to improve chemical reactions—to make them greener and more sustainable—that researchers look for more natural, minimally processed ingredients.
Earwax may not be in sufficient supply to serve as an industrial catalyst, but plenty of other inexpensive, naturally sourced materials are out there, whether they be enzymes derived from pig kidneys or millipede stomachs, metals sequestered by plants, minerals straight from the ground, or metals recovered from highway grit. Here, C&EN takes a look at the prospects of a few “natural catalysts” derived from animal, vegetable, and mineral resources.
Animal: Enzymes from crawling critters
Roger A. Sheldon of Delft University of Technology, an expert in developing enzymes and greener process chemistry for the fine chemicals and pharmaceutical industries, is quick to point out that “enzymes are nature’s sustainable catalysts.” They are derived from renewable resources and are biocompatible, biodegradable, essentially nonhazardous, and nontoxic, he explains. Plus, enzymatic reactions are often performed under mild conditions, avoid the need for scarce precious-metal catalysts, and proceed without waste-generating protection and deprotection steps common in conventional organic syntheses (Green Chem. 2017, DOI: 10.1039/c6gc02157c).
The use of enzymes produced by yeasts dates back thousands of years to bread and cheese making, beer brewing, and wine making, Sheldon says. The first commercial enzyme preparations were produced in the 19th century and included the use of dried calf stomachs in cheese manufacture and animal pancreatic extracts in laundry cleaning. Food and beverage processing together with animal feed and detergents accounts for more than 60% of the enzyme market today, he notes.

However, the development of modern biotechnology in the 1970s brought about a shift from the traditional animal and plant sources, Sheldon says. Genetically modified microorganisms now provide cheaper and higher-purity enzymes that work more efficiently for industrial chemical production.
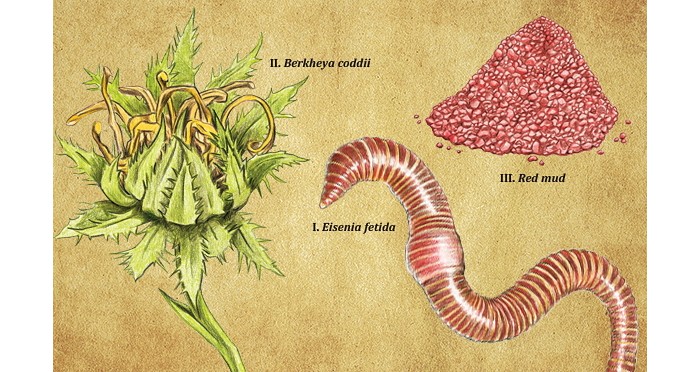
But can chemists come up with enzymes that are even more sustainable? Yan-Hong He and coworkers of Southwest University in Chongqing, China, think so. They have been developing an all-natural catalyst by grinding up earthworms. Like humans, earthworms rely on bacteria in their gut to oxidize and refashion plant and mineral matter to obtain their nutrients. And earthworms can be mass-produced anywhere in compost heaps, living off nothing more than garbage and crop wastes.
Among the enzymes that earthworms exude is a set of serine proteases called earthworm fibrinolytic enzymes. These enzymes are the active ingredients of medicinal extracts used in China for a range of applications, including treating a fever and improving cardiovascular health.
Because enzyme purification can be expensive and time-consuming, He and coworkers began looking for practical ways to use earthworm enzymes directly, without fussing over purifying them or trying to improve their performance through genetic modification. The team was inspired by prior reports of chemists using crude extracts from mushrooms, egg whites, and other sources as biocatalysts.
The researchers rinse common red worms from a worm farm and then puree them in a kitchen blender, He explains. Next, they centrifuge the puddinglike mixture and collect the supernatant liquid, which they dry and then grind into a powder.
The Southwest University chemists first tested the earthworm powder in a Mannich-Michael reaction to prepare isoquinuclidines, which are nitrogen-containing heterocyclic molecules that are useful chemical intermediates. The researchers compared the powder’s abilities with those of commercially available lumbrokinase, one of the earthworm fibrinolytic enzymes, finding that using 100 mg of earthworm powder—the same amount as the purified enzyme—yielded equal results.
They found that their enzyme concoction also works well in asymmetric aldol, Mannich, Henry, and Bignelli reactions (PLOS One 2014, DOI: 10.1371/journal.pone.0105284; Tetrahedron 2015, DOI: 10.1016/j.tet.2015.06.025). Continuing the work, the researchers have just completed the synthesis of the blood-thinning medication warfarin using their earthworm extract.
Natural catalysts provide multiple advantages for chemists, He says. “They are safe, accessible, economical, and environmentally benign.” He acknowledges that the use of the crude enzymes in organic synthesis is limited in terms of the range of reactions that can be catalyzed. “But our work provides a practical way to use sustainable catalysts from nature that could have a transformative effect on streamlining the practice of organic synthesis,” He says.
Yasuhisa Asano of Japan’s Toyama Prefectural University agrees. “Because industrial biotechnology requires more efficient and stable enzymes for sustainable development, we must continuously explore for potential enzyme sources,” Asano says. To that end, Asano’s team has found that an Asian millipede produces a hydroxynitrile lyase enzyme that is much faster at producing cyanohydrins than currently used industrial versions of the enzyme sourced from almonds are (Proc. Natl. Acad. Sci. USA 2015, DOI: 10.1073/pnas.1508311112).
Asano’s team initially collected kilograms of the millipedes to extract the enzyme. But the team studied the enzyme’s amino acid sequence and identified a gene for the enzyme that can be expressed in yeast to ease availability.
Besides working with enzymes from many-legged sources, Asano’s group has also developed a method to make α-aminonitriles and convert them into nonnatural α-amino acids by oxidative cyanation of primary amines using modified enzymes from pig kidneys or the western diamondback rattlesnake (Green Chem. 2017, DOI: 10.1039/c6gc02003h).
“The discovery of millipede hydroxynitrile lyase and the porcine and snake oxidases can serve as a template both for the isolation of promising and efficient enzymes and the design of improved tailor-made enzymes by rational protein engineering,” Asano says.
Vegetable: A bountiful metal harvest
As some researchers have examined metal life cycles—from how the elements are extracted from the ground to how they are recycled or disposed—they’ve started to think about alternative means of acquiring metals that don’t require traditional mining. Their work comes at a time when global ore quality is slipping from high-grade, low-bulk ore to low-grade, high-bulk ore, according to the International Council on Mining & Metals. For example, some economists estimate that we have extracted more commercially accessible metals from the earth than are left in the ground.
An international team of ecologists and metallurgists, including Antony van der Ent and James Vaughan of the University of Queensland and Marie-Odile Simonnot and Guillaume Echevarria of the University of Lorraine, don’t view that situation as a problem. They instead consider it an opportunity. The researchers have been promoting phytoextraction as a sustainable approach to sourcing metals (Environ. Sci. Technol. 2015, DOI: 10.1021/es506031u).
Phytoextraction, also known as phytoremediation or agromining, takes advantage of “hyperaccumulator” plants that have the ability to selectively concentrate high levels of metals from soil or water, van der Ent says. The approach has long been used as a means for cleaning up toxic metals such as arsenic and cadmium at abandoned industrial and mining sites and to prevent the metals from spreading farther in the environment.

Workers typically harvest crops grown on a site, dry and incinerate the biomass to reduce the volume of material, and then bury it in a landfill. But the plant material can also be considered a high-grade “bio-ore” that can be refined into pure metals, van der Ent explains. When you think about the process that way, as agromining, metals can be extracted from mineral-rich soils just about anywhere. Besides providing a source of metals, agromining can help local communities find a use for poor-quality land that isn’t amenable to traditional farming, as well as a source of income, he notes.
Chemists have already turned to using harvested metals as catalysts. Claude Grison and coworkers of France’s CNRS Laboratory of Bioinspired Chemistry & Ecological Innovations (ChimEco) reported in 2012 what is believed to be the first use of hyperaccumulator biomass in organic synthesis (Green Chem. Lett. Rev. 2012, DOI: 10.1080/17518253.2012.667157). The researchers used alpine pennycress grown on a zinc mine site to prepare Lucas’s reagent, a solution of zinc chloride in hydrochloric acid, which they used to make alkyl chlorides from aliphatic alcohols.
The ChimEco process requires a series of steps to isolate crude zinc chloride, including dissolving dried plant matter in acid and ion-exchange chromatography. Still, Grison notes, the process has advantages over purified salts normally used, which “are more expensive and less sustainable to produce.”
Grison and her coworkers continue to expand on the work, using an assortment of hyperaccumulator plants to selectively isolate zinc, nickel, copper, and manganese. They are developing a line of “ecocatalysts” based on the metal salts and have been testing them for possible commercial application in a range of reactions, such as manganese reductive amination of ketones and copper-catalyzed azide-alkyne click chemistry (Green Chem. 2015, DOI: 10.1039/c4gc02193b).
But can chemists come up with ways to use the plant material that are even more sustainable? As Grison and coworkers have shown, crudely isolated metals from plants can be used for some solution-phase reactions. This approach has a limitation, though: Homogeneous catalysis often depends on using a ligand to optimize the electronic and structural properties of a soluble metal complex. But taking a simpler approach and directly using incinerated plant ash as a catalyst is something researchers are starting to think could work.
“Definitely, the metal concentrations in hyperaccumulator plants are so high that specialist chemicals and catalysts can be obtained from them without refining,” van der Ent says. Nickel, which is widely used as a catalyst, can concentrate in hyperaccumulator plants up to about 4% in leaves, he notes, which translates to more than 25% nickel in the resulting ash. That amount compares with only about 2% nickel in low-grade ores.
Preliminary tests by van der Ent and his colleagues have shown that burning hyperaccumulator biomass with a restricted amount of air or reducing the ash with carbon monoxide results in a carbonized material like charcoal that contains nanometer-sized metallic particles. “These particles can be isolated by simple magnetic separation and presumably have excellent catalytic properties,” he says.
In one example, the researchers made carbon-supported metal nanoparticles containing 7% by weight of nickel by pyrolyzing the wood of a tree that grows in New Caledonia. Some 40 years ago, the latex of this tree was shown to contain 25% nickel, the highest known concentration of nickel in a plant. A single tree can contain up to 40 kg of nickel. The team is studying the nanoparticles as a potential hydrogenation catalyst.
The Queensland and Lorraine teams aren’t the first to make nanomaterials from plants partial to a diet of metals. Jiao Qu, Xing Yuan, and coworkers at Northeast Normal University have developed a process to use hyperaccumulator vegetation from a copper-zinc mine to make multiwalled carbon nanotubes, zinc oxide and copper-zinc oxide nanoparticles, and carbon nanotube-metal nanoparticle composites (Environ. Sci.: Nano. 2016, DOI: 10.1039/c6en00287k).
“The nanotubes extracted from hyperaccumulators have shown superior efficiency in electrocatalytic and photocatalytic degradation of organic pollutants in water,” Qu says. Qu believes the metal content is sufficient to use the materials as industrial catalysts as well.
Some researchers have in fact already shown that nanoparticles made from plants are good catalysts. For example, in a proof of concept, a team led by Neil C. Bruce, James H. Clark, and Elizabeth L. Rylott of the University of York grew plants using water spiked with palladium salts and then used the plants to produce catalytic palladium nanoparticles. They have used their heterogeneous plant catalysts in Suzuki-Miyaura coupling reactions to make diaryl compounds. The researchers are now testing the commercial viability of their approach in field trials using plants grown in soil containing mining waste (Environ. Sci. Technol. 2017, DOI: 10.1021/acs.est.6b04821).
Meanwhile, van der Ent and his colleagues continue to explore the possibilities of doing more with less. They’re running a trial in South Africa using a hyperaccumulator aster plant, Burkheya coddii, which is normally used to target nickel, to try to pull palladium and platinum from industrial waste. “The initial results are encouraging, with substantial accumulation of palladium in the plants,” van der Ent says. He points out that all it will take to build the case for the minerals industry to embrace the agromining idea is one large-scale demonstration that works through the operational challenges and provides “real-life evidence of profitability.”
Mineral: Recycling, reusing, and reducing
According to some origin-of-life theories, minerals may have served as the first catalysts of biological chemistry on Earth. Exposed metal ores or minerals known as zeolites and clays might have helped simple molecules coalesce to form biochemical building blocks—amino acids, nucleobases, and sugars.
Chemists today still rely on the catalytic prowess of zeolites, which are porous hydrated aluminosilicates spiked with a variety of metals. For example, zeolite Y is a natural mineral used in catalytic cracking to turn crude oil into useful chemicals, and the synthetic zeolite ZSM-5 is used for hydrocarbon isomerization and alkylation reactions.

“The use of materials based on natural minerals is often the case in industrial catalysis,” says Graham J. Hutchings of Cardiff University, a leading expert in heterogeneous catalysis. In seeking to improve the minerals’ performance, researchers have designed a variety of pretreatment processes, an approach that parallels efforts to improve on enzymes via genetic engineering. For example, Hutchings and his colleagues recently developed a solution-processing method for the mineral malachite to prepare a new class of copper/zinc oxide materials, called zincian georgeite, that is more effective as a catalyst for methanol synthesis and for hydrogen production than currently used malachite catalysts (Chem. Sci. 2017, DOI: 10.1039/c6sc04130b).
But can chemists come up with better uses of mineral resources to make catalysts that are more sustainable? For a growing number of researchers, the answer is yes, and the key is taking advantage of materials that are already out of the ground. Red mud, the noxious by-product of the Bayer process for extracting aluminum from bauxite ore, makes a good case study.
The majority of material processed in mining operations ultimately goes to waste. For every ton of alumina extracted from bauxite, more than a ton of red mud is produced; aluminum mining leaves behind some 120 million metric tons per year of the salty, highly alkaline, heavy-metal-laden material, according to the International Aluminum Institute. Some 4 billion metric tons of the material is lying about globally, much of it held in retention ponds.
Advertisement
Mining companies have long tried to find ways to recycle the environmentally problematic red mud. It is a classic problem in search of a solution. One approach is neutralizing red mud with seawater or treating it with CO2 or sulfur compounds. The modified materials have been tried as fill for mining and construction, as pigment and filler for bricks and cement, and as a sorbent for water treatment. Others have looked at extracting more aluminum from red mud, or obtaining other useful metals such as sodium, copper, and nickel. But so far there have been few safe and economical large-scale applications.
On a new front, some chemists are trying to go catalytic, focusing on iron oxide, the chief component of red mud. But given the purity and properties of red mud, researchers have found it typically is not an active enough catalyst to compete against existing commercial catalysts. That’s because the mineral composition, particle size, and surface properties are important in developing heterogeneous catalysts. With red mud, finding the right combination is a work in progress.
One early sign of success comes from Foster A. Agblevor of Utah State University’s USTAR Bioenergy Center and coworkers in conjunction with Pacific Northwest National Laboratory researchers. They have been testing red mud as a bulk catalyst to replace zeolites in a fluidized-bed reactor to pyrolyze biomass to make crude oil (Energy Fuels 2016, DOI: 10.1021/acs.energyfuels.6b00925). The team processes the biocrude oil using a traditional catalytic hydrotreating process to make a gasoline-type fuel and has tested it on a lawn mower or lawn trimmer. “We are able to run an engine on the fuel without difficulty,” Agblevor says.
The Utah State researchers have applied for a patent for their process. They are working with catalyst company Nexceris to scale up catalyst production and with Wildland Forestry & Environmental to harvest wood from pinyon-juniper range lands in the western U.S. to scale up biofuel production. The team is also expanding the scope of using red mud beyond biomass pyrolysis, Agblevor says. The researchers have applied the catalyst to coal gasification, he notes, as well as to a process for catalytic pyrolysis of waste tires for fuel production.
Despite raw red mud’s ultimate utility as a catalyst, its story points to other possibilities for recovering metals that have already been extracted and used. For example, industrial processing, the use of consumer goods and medicines, and even the wearing away of jewelry leads to measurable amounts of catalyst metals such as gold, silver, and platinum accumulating at wastewater treatment plants.
One of the more prolific sources of these metals, though, is catalytic converters. Automobiles in the U.S. have been using catalytic converters for 40 years. They do a good job on vehicle emissions by zapping pollutants such as unburned hydrocarbons, carbon monoxide, and nitrogen oxides and turning them into more benign products such as CO2, water, and nitrogen.
But as cars putt down the road, catalytic converters slowly disperse platinum, palladium, rhodium, and cerium into the environment. Researchers who have assessed the abundance of these dissipative metals think the concentrations are high enough in the environment, or will be over time, to make it worthwhile to recover them because of their high market values.
To assess the situation, environmental engineer Sebastien Rauch of Chalmers University of Technology and his colleagues measured platinum, palladium, and rhodium concentrations and fluxes in the environment using high-volume air particulate sampling. They have trapped micrometer-sized aerosol particles containing the metals in cities such as Boston and Mexico City and analyzed the filtered samples by inductively coupled plasma mass spectrometry. Concentration ratios of the metals and trace osmium isotope ratios allowed the team to peg catalytic converters as the source of the metals, rather than natural or industrial sources.
Whether one could develop natural catalysts from the errant catalytic converter metals will “depend on the process you want to catalyze and the size of the process,” Rauch says. And harvesting the metals from the air could certainly have some limitations. Consider platinum: A typical automobile exhaust catalyst contains 1 to 2 g of platinum in various combinations with other metals, and the researchers have measured platinum concentrations in urban air of only about 10 pg/m3.
“You can process about 1,500 m3 of air per day using a high-volume sampler,” Rauch says. “After one day, you would only collect about 15 ng. It is of course possible to sample for longer periods, but platinum amounts would remain relatively small.
“Street dust is potentially more interesting,” Rauch continues. Metal concentrations are often in the 100-ng/g (0.1-ppm) range but can be higher at specific locations, he says. “You would need to find a way to concentrate the recovered metals, such as sieving the dust to small particle sizes, so that you could also increase surface area for better contact with the chemicals in the reactions you want to catalyze.”
Testing out the idea, geoscientist Hazel M. Prichard of Cardiff University, who passed away on Jan. 1, became interested in hunting for places where metals tend to accumulate to determine if they are concentrated enough to be worth recovering. Prichard had the idea that recovering the metals could be as simple as scooping up samples from the street or roadsides, storm drains, and wastewater treatment plants. Prichard even investigated the collection bins in the bellies of street-sweeping machines.
Several years ago, Prichard and her colleagues collected samples in Sheffield, England, finding gold primarily from jewelry, and platinum, palladium, and rhodium from catalytic converters. The platinum, palladium, and rhodium combined made up as much as 1.5 ppm of street dust—just at the cusp of what’s needed for catalytic chemistry. They also found more than 3 ppm of gold, platinum, palladium, and rhodium concentrated in incinerated sewage sludge ash.
By comparison, Prichard estimated that the minimum concentration needed to economically extract platinum-group metals from ore deposits is 2–4 ppm. As Rauch points out, mechanical sieving could potentially help concentrate the metals for use as catalysts. As another option, Prichard and her team enlisted biochemist Lynne E. Macaskie of the University of Birmingham to help develop a fermentation process for metal-absorbing microbes to extract metals from the dust. Macaskie and her colleagues have tested palladium-containing bacterial biomass as a bioinorganic catalyst for cleaning up industrial waste and for hydrogenation reactions, finding that the material has potential for industrial applications, she says.
Although recovering metals could prove lucrative, Prichard was motivated by the fact that global supplies of precious metals are limited. Their ongoing use in catalytic converters and handheld electronics could put a crimp on future availability such that some technologies, such as zinc and palladium catalysis, might not be viable long term, and some new technologies, such as fuel-cell cars, won’t be worth pursuing.
“The story of seeking out alternative catalyst sources is an inspiration to the entire chemistry community and elevates the conversation about sustainability to a level at which we can all identify,” says Bruce H. Lipshutz of the University of California, Santa Barbara. Lipshutz and his group have taken a minimalist approach to organic synthesis, using the smallest possible amount of catalyst and organic solvent to see how green and efficient they can make everyday reactions. In one case, they showed that parts-per-million traces of palladium impurity in iron chloride is enough to catalyze cross-coupling reactions.
“Catalysis plays a huge role in minimizing the world’s consumption of precious resources, whether from the mining of metals or reliance on enzymes,” Lipshutz adds. “Claiming both types of catalysts from the earth just makes sense. Finding creative ways to recycle such gifts from nature are clearly important challenges we face, but they are problems we can solve.”




Join the conversation
Contact the reporter
Submit a Letter to the Editor for publication
Engage with us on Twitter