Advertisement
Grab your lab coat. Let's get started
Welcome!
Welcome!
Create an account below to get 6 C&EN articles per month, receive newsletters and more - all free.
It seems this is your first time logging in online. Please enter the following information to continue.
As an ACS member you automatically get access to this site. All we need is few more details to create your reading experience.
Not you? Sign in with a different account.
Not you? Sign in with a different account.
ERROR 1
ERROR 1
ERROR 2
ERROR 2
ERROR 2
ERROR 2
ERROR 2
Password and Confirm password must match.
If you have an ACS member number, please enter it here so we can link this account to your membership. (optional)
ERROR 2
ACS values your privacy. By submitting your information, you are gaining access to C&EN and subscribing to our weekly newsletter. We use the information you provide to make your reading experience better, and we will never sell your data to third party members.
Support nonprofit science journalism
C&EN has made this story and all of its coverage of the coronavirus epidemic freely available during the outbreak to keep the public informed. To support us:
Donate Join Subscribe
Diagnostics
Covid-19
COVID-19 antigen assay set to triple US testing capacity
Abbott aims to produce 50 million of the 15-minute, $5 tests per month
by Mark Peplow, special to C&EN
September 3, 2020
| A version of this story appeared in
Volume 98, Issue 34

A $5 diagnostic test for COVID-19 won emergency authorization from the US Food and Drug Administration (FDA) on August 26 and could see widespread use by the end of this month.
Abbott’s BinaxNOW COVID-19 Ag Card is the first SARS-CoV-2 antigen test to win an emergency use authorization (EUA) that does not require a separate instrument to analyze its result. Instead, the lateral flow assay shows a positive result as a pair of colored lines on a test strip, similar to a pregnancy test, and takes about 15 minutes.
The simplicity of the assay should make it easier to scale up production and deploy it at a large scale. Abbott says it is ready to ship tens of millions of the tests in September, and will increase output to 50 million tests per month in October. The US government has inked a $760 million deal to buy 150 million of the tests, which are likely to be initially deployed in schools, nursing homes, and other areas at high risk of COVID-19 transmission.
The US is currently running about 20 million COVID-19 tests every month, so the new assay could effectively triple national testing capacity. “The goal of tests like this is to give them frequently enough that you can find infections early,” says Rebecca Lee Smith, an epidemiologist at the University of Illinois at Urbana-Champaign.
Assays that use the reverse transcriptase polymerase chain reaction (RT-PCR) to detect RNA from SARS-CoV-2 remain the gold standard for diagnosing COVID-19 infections, but these tests are typically slow and expensive. To overcome this limitation, some universities and other organizations have set up localized sample collection and testing regimes that can return results from saliva-based RT-PCR in a few hours—fast enough to quarantine infected people and their contacts, and limit the spread of disease.
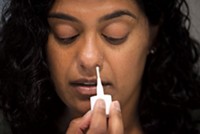
In contrast, BinaxNOW seeks out nucleocapsid proteins from SARS-CoV-2. First, a healthcare worker takes a nasal swab from a patient, and washes the sample onto a test strip inside a credit-card sized mount. As the sample soaks along the strip, antibodies on the strip bind to nucleocapsid protein. This ultimately creates a visible pink/purple line on the strip that indicates a positive result. Other countries have already approved similar instrument-free lateral flow antigen tests, including Pathocatch from Mylab Discovery Solutions in India, and Standard Q from South Korea’s SD Biosensor.
Abbott evaluated its test in 102 patients who had shown COVID-19 symptoms for less than 7 days, and compared the results with a RT-PCR assay. This analysis showed that BinaxNOW has a sensitivity (true positive rate) of 97.1% and a specificity (true negative rate) of 98.5%. The test’s accuracy declined in patients with symptoms that had persisted for more than a week, and the FDA says that negative results in these people should be confirmed with RT-PCR. However, these people are also likely to be less infectious because they typically are carrying smaller viral loads. As such, the test may be well suited to identifying COVID-19 carriers during their first week of symptoms, when they pose the greatest risk of transmitting the disease to others, says virologist Richard S. Tedder at Imperial College London.
BinaxNOW is the fourth antigen test to get an EUA from the FDA. The three previous antigen tests—Quidel’s Sofia; Becton, Dickinson and Company’s Veritor; and the SARS-CoV-2 Ag Test from LumiraDx—all work at a similar speed, but need separate instruments to read the assay’s results with sufficient sensitivity. This requirement has slowed the roll-out of these tests, as manufacturers have struggled to meet demand for the machines.
For now, BinaxNOW tests must be carried out by healthcare professionals in certified labs or authorized point-of-care situations. Patients also must get a prescription for the test. But Abbott has also created a free phone app called NAVICA that will display negative results as a QR code that could serve as a time-limited ‘digital health pass’ to indicate that a person is not contagious. This may suggest that Abbott hopes the test could eventually be deployed more generally for routine screening, says David Grenache, chief scientific officer of TriCore Reference Laboratories in New Mexico, and president of the American Association for Clinical Chemistry.
And more antigen tests are on the horizon. Roche plans to launch its own lateral flow rapid antigen test in late September, and is about to file for an EUA. The company says that its test has a sensitivity of 96.52% and a specificity of 99.68%, based on a trial involving 426 clinical samples, and aims to manufacture 40 million tests per month.
Other companies—including OraSure, E25Bio, and Sona Nanotech—are racing to win authorization for antigen tests that are reliable enough to be self-administered at home. “But in order for home tests to be even remotely useful, there’s going to have to be some requirement around the frequency of testing,” Grenache says.
Smith also worries that if the result of an at-home test determines whether someone can go to work, for example, some people may be tempted to game the system by asking someone else to take the test on their behalf. “We are taking their word that they provided the sample that was tested,” Smith says. “A small group of people behaving badly could sink the whole system.”



Join the conversation
Contact the reporter
Submit a Letter to the Editor for publication
Engage with us on Twitter