Advertisement
Grab your lab coat. Let's get started
Welcome!
Welcome!
Create an account below to get 6 C&EN articles per month, receive newsletters and more - all free.
It seems this is your first time logging in online. Please enter the following information to continue.
As an ACS member you automatically get access to this site. All we need is few more details to create your reading experience.
Not you? Sign in with a different account.
Not you? Sign in with a different account.
ERROR 1
ERROR 1
ERROR 2
ERROR 2
ERROR 2
ERROR 2
ERROR 2
Password and Confirm password must match.
If you have an ACS member number, please enter it here so we can link this account to your membership. (optional)
ERROR 2
ACS values your privacy. By submitting your information, you are gaining access to C&EN and subscribing to our weekly newsletter. We use the information you provide to make your reading experience better, and we will never sell your data to third party members.
Imaging
Color-switching probe clicks onto cell’s molecules
A two-color tag that attaches via click chemistry can track proteins and other cellular components in vivo
by Alla Katsnelson, special to C&EN
February 15, 2019

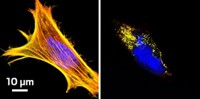
Researchers have developed a fluorescent small molecule that attaches to biomolecules through azide−alkyne cycloaddition—a click reaction—and converts from green to red with the flash of a laser. The molecule, called CPX (clickable and photoconvertible diazaxanthilidene), is less than 500 daltons in size and is also both water soluble and cell permeable, making it a promising probe for tracking proteins and other biomolecules in living cells under a microscope (J. Am. Chem. Soc. 2019, DOI: 10.1021/jacs.8b13094).
Existing probes designed for biological imaging are much bulkier—green fluorescent protein, for example, is 27 kDa in size—and therefore often interfere with the function of the molecules they track. Also, few have CPX’s color-switching capabilities, which allow researchers to track biomolecules such as proteins, lipids, or nucleic acids in more complex ways, says David M. Chenoweth, a chemist at the University of Pennsylvania and the study’s lead author.
The probe’s development stems from a serendipitous discovery that Chenoweth’s lab made while studying the properties of a reported DNA-binding molecule called xylopyridine A, which was isolated by other researchers from a mangrove fungus in 2009. A researcher in Chenoweth’s lab was analyzing the molecule with thin layer chromatography and shined an ultraviolet light on it to visualize it. The molecule started changing from green to red right before his eyes, Chenoweth says, because the light contained the wavelength (440 nm), that initiates the photoconversion.

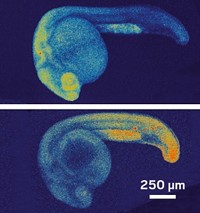
The team realized that this color-switching property would make it possible to track biomolecules as they perform specific functions in vivo, Chenoweth says.
To make the molecule easy to attach to biomolecules, Chenoweth and his team added a clickable azide handle to one of its pyridine nitrogens. This allows the molecule to attach via a cycloaddition reaction between the azide of the dye and an alkyne in a biomolecule—which can be easily introduced through synthesis, semisynthesis, enzymatic, or bioengineering techniques.
Then they tested the molecule in two proof-of-principle experiments. First, they attached the molecule to a lipid and found that it didn’t interfere with the lipid’s ability to self-assemble into vesicles. The vesicles fluoresced and were easy to see under the microscope. The researchers could also shine UV light on select vesicles and change their color, which gave the researchers the ability to track the movement of specific subsets of vesicles.
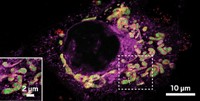
In the second experiment, they attached the probe to a protein called α-synuclein, which aggregates in neurons in Parkinson’s disease. Working with collaborators who bioengineered the protein to contain an alkyne onto which the probe could click, they showed that having the probe attached did not interfere with α-synuclein’s ability to aggregate. After attaching the probe, they could follow the protein inside neurons and photoswitch just the ones that aggregated, allowing them to study how aggregates formed in more detail. “I think this tool is really primed for use in investigating the dynamics of α-synuclein and other molecules in a spatially and temporally defined way,” Chenoweth says.
Advertisement
Because the CPX molecule has several aromatic rings, the researchers envision being able to tune the color of the fluorescence by putting electron-donating or electron-withdrawing groups onto individual rings. They are also working on modifying the structure of the molecule to open up the possibility of switching among more than two colors, which would allow researchers to monitor multiple related biomolecules with just one probe.
“The paper is a very, very nice piece of work. We want to use this thing, because it’s really quite cool,” says Carston Wagner, a chemical biologist at the University of Minnesota. “I think it has the potential to be a really widely used probe."


Join the conversation
Contact the reporter
Submit a Letter to the Editor for publication
Engage with us on Twitter