Advertisement
Grab your lab coat. Let's get started
Welcome!
Welcome!
Create an account below to get 6 C&EN articles per month, receive newsletters and more - all free.
It seems this is your first time logging in online. Please enter the following information to continue.
As an ACS member you automatically get access to this site. All we need is few more details to create your reading experience.
Not you? Sign in with a different account.
Not you? Sign in with a different account.
ERROR 1
ERROR 1
ERROR 2
ERROR 2
ERROR 2
ERROR 2
ERROR 2
Password and Confirm password must match.
If you have an ACS member number, please enter it here so we can link this account to your membership. (optional)
ERROR 2
ACS values your privacy. By submitting your information, you are gaining access to C&EN and subscribing to our weekly newsletter. We use the information you provide to make your reading experience better, and we will never sell your data to third party members.
Imaging
Rigid tip improves atomic force microscopy imaging
An inflexible copper oxide AFM tip allows researchers to observe hydrogen bonds between molecules
by Sam Lemonick
April 12, 2018
| A version of this story appeared in
Volume 96, Issue 16

Atomic force microscopy (AFM) can get chemists pretty close to actually seeing the atoms and bonds of molecules. The technique produces images a bit like a record player generates sound. An atomic-scale needle at the end of a cantilevered arm scans across a sample, moving up and down according to the shape and electronic properties of the surface. Measuring and recording that deflection produces a three-dimensional representation of the molecules within the sample.

Minimizing interactions between the needle and the sample can lead to sharper images. In 2009, IBM researchers proposed using a carbon monoxide tip to significantly improve AFM resolution. The drawback is that atoms on a sample’s surface can tug on the CO dangling from the end of a metal needle, making bonds look longer than they actually are or creating artifacts that look like bonds between atoms.
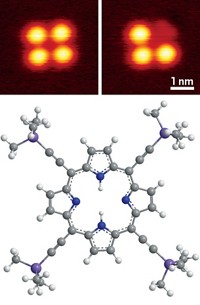
Now, researchers have swapped this flexible CO tip for a more rigid one to improve AFM’s ability to image inter- and intramolecular bonds (Nat. Nanotechnol. 2018, DOI: 10.1038/s41565-018-0104-4).
Taking inspiration from a 2012 paper in Physical Review B that proposed using a copper oxide tip for AFM, Harry Mönig of the University of Münster and colleagues demonstrated that such a needle can reliably image bonds without false positives and can accurately measure bond lengths. The copper oxide tip is about one order of magnitude more rigid than its CO counterpart.
“This is a very significant paper,” says Xiaohui Qiu, a physical chemist at the National Center for Nanoscience & Technology, explaining that the work represents a significant advance in noncontact AFM imaging. Qiu says Mönig’s paper resolves a debate that has been ongoing since 2013 about whether hydrogen bonds in AFM images are real or artifacts, and the work shows that AFM can reliably characterize both inter- and intramolecular bonds.
Mönig’s group had previously shown that their copper oxide-tipped needle could be used in AFM. In the new paper, they first demonstrate the tip’s ability to accurately measure bond length by scanning a buckyball. The bond lengths in the AFM images were within two tenths of an angstrom of the bond lengths obtained by X-ray diffraction and predicted by density functional theory. CO tips overestimate these bond lengths by as much as an angstrom. The stiffer copper oxide tips did sacrifice some sharpness in imaging relative to CO tips.
The researchers also successfully imaged a predicted nitrogen-gold-nitrogen bond between bis(para-pyridyl)acetylene and a gold surface. Chemists had debated the existence of this bond because of discrepancies between simulations and AFM images made with a CO tip.
Advertisement
Finally, the group imaged intermolecular hydrogen bonding between 3,4,9,10-perylene-tetracarboxylic-dianhydride (PTCDA) molecules. The flexibility of CO tips can produce lines of contrast that look like bonds between atoms in AFM images, making it hard to distinguish between actual hydrogen bonds and artifacts. Mönig’s group found that copper oxide tips did not produce those kinds of artifacts in the PTCDA images.
To make these new rigid tips, Mönig’s team first presses a blunt tungsten needle into a copper surface to pick up a pyramid of copper atoms. They then dip it just tenths of angstroms deep in a copper oxide surface. The process reliably adds copper oxide to the tip, although Mönig says it can be challenging to make sure the copper oxide pyramid has one oxygen aiming straight down.



Join the conversation
Contact the reporter
Submit a Letter to the Editor for publication
Engage with us on Twitter