Advertisement
Grab your lab coat. Let's get started
Welcome!
Welcome!
Create an account below to get 6 C&EN articles per month, receive newsletters and more - all free.
It seems this is your first time logging in online. Please enter the following information to continue.
As an ACS member you automatically get access to this site. All we need is few more details to create your reading experience.
Not you? Sign in with a different account.
Not you? Sign in with a different account.
ERROR 1
ERROR 1
ERROR 2
ERROR 2
ERROR 2
ERROR 2
ERROR 2
Password and Confirm password must match.
If you have an ACS member number, please enter it here so we can link this account to your membership. (optional)
ERROR 2
ACS values your privacy. By submitting your information, you are gaining access to C&EN and subscribing to our weekly newsletter. We use the information you provide to make your reading experience better, and we will never sell your data to third party members.
Biological Chemistry
Synthetic Route to Tetracyclines
Modular, flexible synthesis yields structurally diverse antibiotics
by Amanda Yarnell
April 18, 2005
| A version of this story appeared in
Volume 83, Issue 16

ORGANIC CHEMISTRY
A practical synthetic route developed by researchers at Harvard University has yielded an unprecedented series of structurally diverse analogs of the antibiotic tetracycline, including those capable of killing bacteria that are resistant to multiple antibiotics (Science 2005, 308, 395).
Tetracycline was once an enormously effective treatment for a wide range of bacterial infections, but widespread resistance against the antibiotic has developed. Because no practical synthetic route to this complex antibiotic is available, the quest for better performing analogs has been restricted to semisynthesis, whereby the natural product is isolated and chemically modified, Harvard chemistry professor Andrew G. Myers says.
"This approach greatly limits the range of accessible structures that might be studied as new antibiotic candidates," Myers points out. Now, after more than a decade of work, he and his coworkers--including postdoc Christian D. Lerner and graduate students Mark G. Charest, Jason D. Brubaker, and Dionicio R. Siegel--have devised a flexible and modular route to a diverse range of tetracycline analogs.
This route "is not just a synthetic tour de force; it enormously expands the armamentarium of the tetracycline medicinal chemist," note Chaitan Khosla of Stanford University and Yi Tang of the University of California, Los Angeles, in an accompanying commentary.
Tetracycline consists of four linearly fused rings, designated A, B, C, and D. Whereas previous synthetic attempts have involved stepwise assembly of the ABCD-ring system, the Harvard team constructs an AB-ring precursor enone and a D-ring precursor carbanion, then joins them via the Michael-Dieckmann reaction to yield an ABCD-ring tetracycline. In principle, this reaction could have yielded a mixture of four tetracycline stereoisomers. Instead, the analogs are formed as largely one diastereomer with stereochemistry that matches that of the natural product.

Gilbert Stork of Columbia University, whose lab has also wrestled with the synthesis of tetracycline, notes that Myers' "versatile, general synthesis is a beautiful solution to a difficult problem."
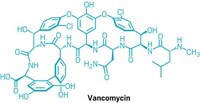
Tetracycline exerts its antibacterial effect by binding to the small (30S) subunit of the bacterial ribosome. Bacteria have figured out various ways to outsmart the antibiotic, including producing a protein that purges the ribosome of tetracycline or one that pumps the antibiotic out of the cell. X-ray crystallography has revealed that certain regions of tetracycline make extensive contacts with the ribosome. It has been suggested that tetracycline analogs with modifications outside these regions might be able to overcome bacterial resistance, Myers notes. Indeed, one of his team's analogs--one with a fifth ring fused to the D-ring of tetracycline--is active against a range of gram-positive bacteria, including strains resistant to tetracycline, methicillin, and vancomycin.
Myers points out that, although yields remain too low (5–8%) for the current route to be competitive with commercial semisynthetic preparations of tetracycline analogs, a range of analogs not available by semisynthesis can now be rapidly prepared in amounts sufficient for antibacterial evaluation.



Join the conversation
Contact the reporter
Submit a Letter to the Editor for publication
Engage with us on Twitter