Advertisement
Grab your lab coat. Let's get started
Welcome!
Welcome!
Create an account below to get 6 C&EN articles per month, receive newsletters and more - all free.
It seems this is your first time logging in online. Please enter the following information to continue.
As an ACS member you automatically get access to this site. All we need is few more details to create your reading experience.
Not you? Sign in with a different account.
Not you? Sign in with a different account.
ERROR 1
ERROR 1
ERROR 2
ERROR 2
ERROR 2
ERROR 2
ERROR 2
Password and Confirm password must match.
If you have an ACS member number, please enter it here so we can link this account to your membership. (optional)
ERROR 2
ACS values your privacy. By submitting your information, you are gaining access to C&EN and subscribing to our weekly newsletter. We use the information you provide to make your reading experience better, and we will never sell your data to third party members.
Synthesis
'Ring-walking' Metals Yield Clues about Catalysis
ACS MEETING NEWS
by Stephen K. Ritter
September 19, 2005
| A version of this story appeared in
Volume 83, Issue 38

For some time, chemists have considered the possibility that once a transition metal coordinates to an olefin or aromatic ring, the metal center might then migrate sequentially to other parts of the molecule, forming a series of intermediates along the way before finding a favored spot to react. This "ring-walking" phenomenon is thought to be a key process in the metal-mediated activation of carbon-hydrogen and carbon-halogen bonds.
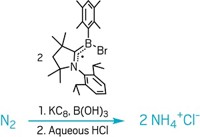
A model reaction system introduced by Milko E. van der Boom of Weizmann Institute of Science, in Rehovot, Israel, and his coworkers may now make it easier for chemists to explore the ring-walking mechanism. The ability to zoom in and visualize if ring walking occurs or not in step-by-step detail could be very useful in helping to design catalysts and reaction systems that have better selectivity and are more efficient.
"Ring walking is often difficult to prove unambiguously, as the vast majority of metal-promoted arene functionalization reactions are bimolecular, require elevated temperatures, and the intermediates are not observed," van der Boom noted.
His group has turned to a model set of electron-rich 1⁄4-conjugated systems and is using nuclear magnetic resonance spectroscopy experiments to track formation of intermediate species under varying reaction conditions. The work is supported by computational studies carried out by the group of Weizmann colleague Jan M. L. Martin.
The researchers studied room-temperature coordination of a platinum phosphine complex to the double bond of a brominated stilbazole, a compound in which a bromophenyl ring is joined to a pyridine ring via an ethylene bridge (J. Am. Chem. Soc. 2005, 127, 9322). Running the same reaction at 65 °C leads to formation of a compound in which the platinum complex instead adds to the C-Br bond of the phenyl ring. They also showed that the olefin-coordinated complex can be converted into the addition product simply by heating the solid or a solution of the olefin complex. The researchers have extended the work to include nickel and palladium phosphine analogs, with similar results.
The team members deduced that the activation barrier for olefin coordination in their stilbazole system is lower than that for the addition reaction. They concluded that metal-olefin dissociation doesn't occur under heating but that the metal center proceeds to walk over the 1/4-conjugated system, forming transient intermediates along the way until it reaches and activates the C-Br bond in a unimolecular process. These findings are in contrast to what might be expected for typical organometallic transformations in which the metal-olefin complex would dissociate and aryl-halogen activation would subsequently occur in a bimolecular process without ring walking, van der Boom said.
The Weizmann researchers are in the process of determining whether ring walking is a general phenomenon and how fast or how far a metal center might be able to travel. For example, van der Boom described an azostilbazole system in which the central alkene (C=C) was replaced by an azo (N=N) group. Another example is a symmetrical three-ring system consisting of a central phenyl ring connected to pyridine rings on either side by ethylene bridges. In both cases, they observed the same initial coordination to the double bond followed by ring walking and then addition. The team also is exploring the design of molecules in which the 1/4-conjugation is broken in order to control the direction of the ring walking.
"Ring walking may be of practical and synthetic value, as it could be used to control the regio- and stereoselectivity of metal-mediated transformations and homogeneous catalysis," van der Boom concluded.


Join the conversation
Contact the reporter
Submit a Letter to the Editor for publication
Engage with us on Twitter