Advertisement
Grab your lab coat. Let's get started
Welcome!
Welcome!
Create an account below to get 6 C&EN articles per month, receive newsletters and more - all free.
It seems this is your first time logging in online. Please enter the following information to continue.
As an ACS member you automatically get access to this site. All we need is few more details to create your reading experience.
Not you? Sign in with a different account.
Not you? Sign in with a different account.
ERROR 1
ERROR 1
ERROR 2
ERROR 2
ERROR 2
ERROR 2
ERROR 2
Password and Confirm password must match.
If you have an ACS member number, please enter it here so we can link this account to your membership. (optional)
ERROR 2
ACS values your privacy. By submitting your information, you are gaining access to C&EN and subscribing to our weekly newsletter. We use the information you provide to make your reading experience better, and we will never sell your data to third party members.
Environment
CO2 enables 'switchable surfactants'
August 21, 2006
| A version of this story appeared in
Volume 84, Issue 34
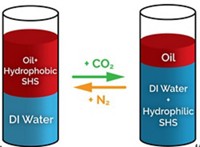
Being able to reversibly generate stable emulsions could help facilitate syntheses, separations, and processing of a variety of chemicals. Philip G. Jessop of Queen's University, Kingston, Ontario, and his colleagues have made this possible by creating "switchable surfactants" that need only CO2 and air to turn on and off (Science 2006, 313, 958). A year ago, Jessop's group reported switchable solvent systems based on the same concept (C&EN, Aug. 29, 2005, page 27). The researchers used long-chain alkylamidines RN=CR'-NR'2, where R is C12H25 or C16H33 and R' is methyl—that form bicarbonate salts when mixed with water and CO2 is added. The in situ salt formation helps create stable emulsions. Bubbling air into the mixture releases CO2 and breaks the emulsion. Switchable surfactants can be considered for all process applications involving surfactants, including emulsion polymerizations and different aspects of crude oil processing, Jessop notes.



Join the conversation
Contact the reporter
Submit a Letter to the Editor for publication
Engage with us on Twitter