Advertisement
Grab your lab coat. Let's get started
Welcome!
Welcome!
Create an account below to get 6 C&EN articles per month, receive newsletters and more - all free.
It seems this is your first time logging in online. Please enter the following information to continue.
As an ACS member you automatically get access to this site. All we need is few more details to create your reading experience.
Not you? Sign in with a different account.
Not you? Sign in with a different account.
ERROR 1
ERROR 1
ERROR 2
ERROR 2
ERROR 2
ERROR 2
ERROR 2
Password and Confirm password must match.
If you have an ACS member number, please enter it here so we can link this account to your membership. (optional)
ERROR 2
ACS values your privacy. By submitting your information, you are gaining access to C&EN and subscribing to our weekly newsletter. We use the information you provide to make your reading experience better, and we will never sell your data to third party members.
Analytical Chemistry
Tying Up Loose Ends, Chemically Speaking
by Bethany Halford
September 25, 2006
| A version of this story appeared in
Volume 84, Issue 39
At present, the practical applications of molecular knots are arguably few, but the challenge of creating such structures has intrigued synthetic chemists for years. The mental gymnastics it takes to tie the molecules is also a good way to get fledgling organic chemists to appreciate molecules in 3-D. Edward E. Fenlon, a chemistry professor at Franklin & Marshall College, in Lancaster, Pa., has been successful at snagging a few bright undergrads to conduct research on molecular knots.
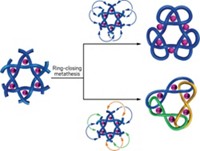
Fenlon spoke about the progress he and undergraduate Matt Dietz have made toward preparing the world's smallest trefoil knot during a session sponsored by the Division of Organic Chemistry. Trefoil knots possess three crossings, making them the simplest of all knots. The smallest molecular trefoil knots deliberately synthesized to date have at least 80 backbone atoms.
Fenlon and Dietz have been working toward a C63H126 knot. If successful, they will have created the smallest knot yet and the only purely hydrocarbon knot ever prepared. The critical step uses olefin metathesis to intramolecularly link together the trefoil precursor's six arms. Two threading events must take place at a specific stage of the metathesis in order to tie the knot.
Upon hydrolysis of a key ester, Fenlon and Dietz were able to isolate three large triols—each of the same molecular weight—using preparatory thin-layer chromatography. Fenlon thinks these are a large macrocycle, a catenane, and the trefoil. Although preliminary analysis looks promising, Fenlon told C&EN he'd like to get unambiguous proof, such as an X-ray crystal structure, before declaring victory over the knot. He and Dietz are currently trying to derivatize the structure so it's more likely to crystallize.



Join the conversation
Contact the reporter
Submit a Letter to the Editor for publication
Engage with us on Twitter