Advertisement
Grab your lab coat. Let's get started
Welcome!
Welcome!
Create an account below to get 6 C&EN articles per month, receive newsletters and more - all free.
It seems this is your first time logging in online. Please enter the following information to continue.
As an ACS member you automatically get access to this site. All we need is few more details to create your reading experience.
Not you? Sign in with a different account.
Not you? Sign in with a different account.
ERROR 1
ERROR 1
ERROR 2
ERROR 2
ERROR 2
ERROR 2
ERROR 2
Password and Confirm password must match.
If you have an ACS member number, please enter it here so we can link this account to your membership. (optional)
ERROR 2
ACS values your privacy. By submitting your information, you are gaining access to C&EN and subscribing to our weekly newsletter. We use the information you provide to make your reading experience better, and we will never sell your data to third party members.
Synthesis
'Sulflower' circulene debuts
October 23, 2006
| A version of this story appeared in
Volume 84, Issue 43
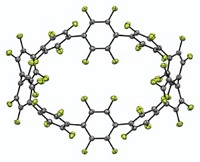
Sometimes a molecule's name is inspiration enough to instigate its synthesis. But the molecule nicknamed "sulflower"—a mash-up of sulfur and flower—recently synthesized by a team in Russia, has more going for it than a clever moniker (Angew. Chem. Int. Ed., DOI: 10.1002/anie.200602190). The compound (shown), also known as octathio[8]circulene, (C2S)8, represents a new form of carbon sulfide, according to Valentine G. Nenajdenko and colleagues at Moscow State University. Sulflower is the first fully heterocyclic circulene, and it may possess electronic properties similar to those of other oligothiophenes, which are popular materials in light-emitting devices and thin-film transistors. The researchers started from an eight-membered carbon ring symmetrically fused with four thiophene moieties. They then sulfurized the unsubstituted carbon atoms of the thiophene moieties, and acidification and vacuum pyrolysis knit the acyclic sulfur atoms of this intermediate into the circulene. By varying the heteroatoms, Nenajdenko hopes to make other novel heterocyclic circulenes.




Join the conversation
Contact the reporter
Submit a Letter to the Editor for publication
Engage with us on Twitter