Advertisement
Grab your lab coat. Let's get started
Welcome!
Welcome!
Create an account below to get 6 C&EN articles per month, receive newsletters and more - all free.
It seems this is your first time logging in online. Please enter the following information to continue.
As an ACS member you automatically get access to this site. All we need is few more details to create your reading experience.
Not you? Sign in with a different account.
Not you? Sign in with a different account.
ERROR 1
ERROR 1
ERROR 2
ERROR 2
ERROR 2
ERROR 2
ERROR 2
Password and Confirm password must match.
If you have an ACS member number, please enter it here so we can link this account to your membership. (optional)
ERROR 2
ACS values your privacy. By submitting your information, you are gaining access to C&EN and subscribing to our weekly newsletter. We use the information you provide to make your reading experience better, and we will never sell your data to third party members.
Biological Chemistry
Carbon Nanotubes Pass Safety Test
Functionalized carbon nanotubes are rapidly cleared from blood, excreted in urine
by Michael Freemantle
February 14, 2006

A new study could allay some of the fears about health and safety issues relating to carbon nanotubes and open the door for exploring the use of such nanomaterials in the body.

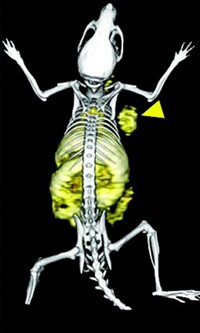
In the study, mice were intravenously administered water-soluble carbon nanotubes, either single-walled or multiwalled. Electron microscopy analysis revealed that both types of nanotubes are excreted intact in urine.
The work was carried out at the Centre for Drug Delivery Research at the University of London's School of Pharmacy and was led by Kostas Kostarelos, deputy head of the center, in collaboration with organic chemists Alberto Bianco at the CNRS Institute of Molecular & Cellular Biology, in Strasbourg, France, and Maurizio Prato of the University of Trieste, in Italy (Proc. Natl. Acad. Sci. USA, published online, dx.doi.org/10.1073/pnas.0509009103).
"This is the first time carbon nanotubes have been administered intravenously and fundamental pharmacokinetic parameters have been obtained," Kostarelos tells C&EN. "It is also the first description of carbon nanotubes circulating in the blood of live animals and the first report showing blood clearance and urine excretion of the nanotubes."
From the point of view of biomedical applications, identifying the pharmacological limitations and advantages of such nanostructures is imperative for any kind of therapeutic or diagnostic application that involves administering or implanting carbon nanotubes in patients, he notes.
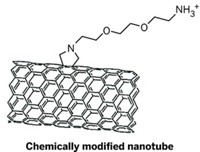
The researchers solubilized the nanotubes by functionalizing them with hydrophilic amino groups, using 1,3-dipolar cycloaddition of azomethine ylides. They then covalently linked the amino groups to a diethylenetriaminepentaacetic chelating agent. The radioactive isotope indium-111 complexed with the agent was used for imaging the nanotubes.
Radioactivity tracing showed that the intravenously administered soluble single-walled nanotubes were not retained in organs such as the heart, liver, and spleen. The team did not investigate the tissue biodistribution and blood clearance rates of the multiwalled tubes but carried out urine excretion studies on both types of tubes, using transmission electron microscopy.
"This careful study" has dismissed many of the worries of using nanotubes in therapeutics, showing that solubilized nanotubes "flush through [mice] without being retained in organs," says carbon nanotube expert Pulickel M. Ajayan, professor of engineering at Rensselaer Polytechnic Institute, Troy, N.Y. "This certainly removes the shroud that has made many people nervous about using nanotubes, especially for medical applications, and opens the way for more dedicated work to develop nanotube-based therapeutics."


Join the conversation
Contact the reporter
Submit a Letter to the Editor for publication
Engage with us on Twitter