Advertisement
Grab your lab coat. Let's get started
Welcome!
Welcome!
Create an account below to get 6 C&EN articles per month, receive newsletters and more - all free.
It seems this is your first time logging in online. Please enter the following information to continue.
As an ACS member you automatically get access to this site. All we need is few more details to create your reading experience.
Not you? Sign in with a different account.
Not you? Sign in with a different account.
ERROR 1
ERROR 1
ERROR 2
ERROR 2
ERROR 2
ERROR 2
ERROR 2
Password and Confirm password must match.
If you have an ACS member number, please enter it here so we can link this account to your membership. (optional)
ERROR 2
ACS values your privacy. By submitting your information, you are gaining access to C&EN and subscribing to our weekly newsletter. We use the information you provide to make your reading experience better, and we will never sell your data to third party members.
Biological Chemistry
Chemistry, Biology Cross-Fertilization
Young field provides new tools for biology, new applications for chemistry
by Stu Borman
October 1, 2007
| A version of this story appeared in
Volume 85, Issue 40

"CHEMICAL BIOLOGY is an exploding field that has opened up the use of chemistry by many molecular and cell biologists who would otherwise not now be using it to help understand biological systems," and it has also provided a wider range of applications for research carried out by chemists, said Giulio Superti-Furga, director of the Research Center for Molecular Medicine of the Austrian Academy of Sciences, in Vienna.
To advance the field of chemical biology, Superti-Furga and assistant professor Ulrike Eggert of the Dana-Farber Cancer Institute and the department of biological chemistry and molecular pharmacology at Harvard Medical School recently organized a European Molecular Biology Organization workshop on "Drug Action & Chemical Biology in the Post-Genomic Era." The workshop was held in August at the 18th-century headquarters of the Austrian Academy of Sciences, in a section of Vienna where the narrow cobblestone streets date back to medieval times.
The meeting took place in the academy building's ornate Festive Hall, which has a huge baroque fresco on its ceiling and statues gracing nooks along the walls. In the early 1800s, musicians Franz Joseph Haydn, Ludwig van Beethoven, Franz Schubert, and Antonio Salieri performed there.
Now, scientists were there to discuss how chemists and biologists have increasingly been making beautiful music together in their efforts to learn more about the chemistry of biological systems and to discover new drugs. Eggert and Superti-Furga noted that the meeting aimed "to bridge cultures and allow cross-fertilization of disciplines."
CHEMICAL BIOLOGY is a relatively new field that began to attract attention and gain popularity only in the early 1990s. Several workshop participants said they believed the term "chemical biology" was invented by Harvard professor of chemistry and chemical biology Stuart L. Schreiber, who cofounded the first journal in the field, Chemistry & Biology. Schreiber, who was invited to the Vienna workshop but was unable to attend, may indeed have helped popularize the term but says he didn't invent it.
"I don't know its origins, but I recall vividly [the late biochemist and Nobel Prize winner] Konrad E. Bloch using it when I was a graduate student at Harvard in the '70s," Schreiber says. "I think the meaning behind the term has evolved with time, but that has been a gradual process, with no one paper or lecture, to my knowledge, being the basis for its widespread use today."
Some researchers, including some workshop participants, are still uncertain about how the field is defined and how it differs from existing disciplines like biochemistry and molecular biology. Eggert said she likes to define chemical biology "in broad terms, as the use of tools and approaches of chemistry to understand aspects of biology." She noted that Harvard's chemical biology graduate program defines it as a field that "differs from chemistry because one of its major goals is to understand biology and differs from biology because it predominantly uses approaches from chemistry."
There indeed are clear distinctions between chemical biology and similar-sounding fields. Biochemistry is the study of the way biomolecules act in cells and organisms, and molecular biology "focuses on the macromolecules—DNA, RNA, and proteins—that enable heritable-information flow" in cells, organs, and organisms, Schreiber says, whereas chemical biology explores interactions of small molecules with such macromolecules. The key to chemical biology is thus its use of small molecules to perturb, poke, and probe biological systems.
Eggert and workshop attendee Matthew Bogyo both pointed out that chemical biology is closer to pharmacology than it is to biochemistry and molecular biology. Like chemical biology, pharmacology "also puts the focus on small molecules," said Bogyo, assistant professor of pathology at Stanford University School of Medicine. However, pharmacology deals with the influence of drugs on biological systems, whereas "chemical biology tends to be more global in its approach," he noted, focusing not only on drugs but also on nondrug small molecules.
In his workshop presentation, Bogyo discussed his own group's recent development of nondrug small molecules. In the study, he and his coworkers developed small-molecule fluorescent probes that can monitor the presence and bioactivity of key enzymes (Nat. Chem. Biol. 2007, 3, 668). The probes are potentially useful to diagnose disease and to test for key properties of small-molecule therapeutics in vivo.
MANY OTHER studies discussed at the Vienna meeting exemplify the interface between chemical biology and drug discovery. For example, professor of chemistry and biochemistry Ronald T. Raines of the University of Wisconsin, Madison, described how his group modified the sequence of the human RNase 1 enzyme to create variants that are preferentially toxic to cancer cells. Quintessence Biosciences, a biopharmaceutical company in Madison that Raines cofounded, is planning to conduct a Phase I clinical trial of one of these RNase 1 variants for treatment of a variety of cancers in early 2008. Raines noted that he and his coworkers "are now using a variety of tools, especially new small-molecule probes known as 'fluorogenic labels,' to delineate the mechanism by which our cytotoxic ribonucleases kill cancer cells." They are also trying to extend their enzyme-modification concept to other diseases and to antiviral applications by creating synthetic zymogens, inactive enzyme precursors that can be activated by disease-specific enzymes in vivo.

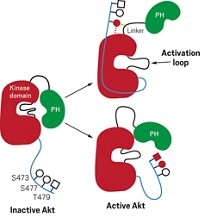
Nathanael S. Gray of Dana-Farber Cancer Institute and the department of biological chemistry and molecular pharmacology at Harvard Medical School discussed his group's recent screening of a combinatorial heterocycle library for inhibitors of Bcr-abl, a tyrosine kinase encoded by an abnormal chromosome found in about 95% of people with chronic myelogenous leukemia (Nat. Chem. Biol. 2006, 2, 95). Compounds identified in the study, including a lead compound called GNF-2, show anticancer activity exclusively against cells bearing the Bcr-abl mutation.
Such specificity is difficult to achieve with tyrosine kinase inhibitors, since many types of tyrosine kinases are found throughout the body. GNF-2 achieves specificity because the spot where it binds to its enzyme target and its allosteric mechanism of action are both unusual. An allosteric mechanism is one caused by an agent that binds an enzyme at a location outside the enzyme's active site. In 2003, Superti-Furga, John Kuriyan of Howard Hughes Medical Institute and the department of molecular and cell biology at the University of California, Berkeley, and coworkers first identified and described the same unusual binding pocket and suggested its use as a drug target.
Péter Lénárt, a postdoc in the group of senior scientist Jan-Michael Peters at the Research Institute of Molecular Pathology, Vienna, discussed a study in which a small-molecule inhibitor was used to identify the role a kinase called Plk1 plays in mitosis. "I was primarily interested in the biology, but in the end, the results were also of interest to Boehringer Ingelheim, which provided the inhibitor and is currently investigating it in clinical trials for a broad range of cancer types," Lénárt said.
Stephen W. Fesik, divisional vice president of cancer research at Abbott Laboratories, Abbott Park, Ill., described over a decade of research on the Bcl-2 family of proteins, which play key roles in apoptosis (cell suicide). He and his coworkers identified small-molecule inhibitors of Bcl-2 proteins by using "SAR by NMR" (a fragment-based drug discovery technique they invented in 1996) as well as structure-based drug design, parallel synthesis, and medicinal chemistry. One of the agents they identified, ABT-263, is currently in Phase I clinical trials for lymphoma, small-cell lung cancer, and chronic lymphocytic leukemia.
In addition to yielding this promising drug candidate, Abbott's chemical biology studies on Bcl-2 proteins also might lead to a better fundamental understanding of apoptotic signal transduction and the role of apoptosis in cancer. "How critical are individual Bcl-2 family members for cancer cell viability?" Fesik asked. "If we disrupt the natural process by which Bcl-2 family members inhibit programmed cell death, will we kill cancer cells? Do cancer cells depend on Bcl-2 more than normal cells do, and can we therefore get a therapeutic window? We've given out Bcl-2 inhibitor probes to over 75 laboratories to promote such studies."
Fesik noted that in the pharmaceutical industry, collaboration between chemists and biologists has proven essential for successful drug discovery. "At Abbott, we are organized by project teams, each of which has a chemistry group and a biology group that are closely linked together," he said. "In the past, collaborations between chemists and biologists in academia have not been as strong," but he believes the advent of chemical biology has resulted in a growing number of such cooperative efforts.
"IN GENERAL, I think the field of chemical biology is progressing nicely," Bogyo said. "There has been an increase in the number of medical schools and basic science campuses with a chemical biology focus both in course work and in graduate programs, and there are a number of new chemical biology-focused journals, including ACS Chemical Biology and Nature Chemical Biology . I see the future of chemical biology not necessarily in the connections or collaborations between pure chemical and pure biological labs, but rather in the growing number of chemists who want to think about biological problems and of biologists who can do enough chemistry to make compounds that advance their biological research."
Eggert noted that "Giulio and I are writing an in-depth discussion of the science discussed at the chemical biology workshop in a meeting report that will come out in the January issue of Nature Chemical Biology."
Some might consider "chemical biology" to be just a new name, she added. "But giving the field a new name got people more excited about using chemistry to interrogate biological questions, and that's not a bad thing."



Join the conversation
Contact the reporter
Submit a Letter to the Editor for publication
Engage with us on Twitter