Advertisement
Grab your lab coat. Let's get started
Welcome!
Welcome!
Create an account below to get 6 C&EN articles per month, receive newsletters and more - all free.
It seems this is your first time logging in online. Please enter the following information to continue.
As an ACS member you automatically get access to this site. All we need is few more details to create your reading experience.
Not you? Sign in with a different account.
Not you? Sign in with a different account.
ERROR 1
ERROR 1
ERROR 2
ERROR 2
ERROR 2
ERROR 2
ERROR 2
Password and Confirm password must match.
If you have an ACS member number, please enter it here so we can link this account to your membership. (optional)
ERROR 2
ACS values your privacy. By submitting your information, you are gaining access to C&EN and subscribing to our weekly newsletter. We use the information you provide to make your reading experience better, and we will never sell your data to third party members.
Synthesis
ACS, Pharma Initiative Awards Additional Grant
Joint effort targets more efficient Suzuki C???C coupling reactions that partially avoid halogenated aromatics
by Stephen K. Ritter
July 11, 2007

The Pharmaceutical Roundtable of the Green Chemistry Institute, a division of the American Chemical Society, has awarded a second research grant as part of a broad effort to advance green chemistry and engineering practices in the global pharmaceutical industry.

The $100,000 award goes to chemistry professors Robert E. Maleczka Jr. and Milton R. Smith III of Michigan State University. Half of the funds were provided by the roundtable and half by Pfizer, one of the roundtable members. Maleczka and Smith will use the grant to continue their work on Suzuki-type C???H activation and C???C coupling of aromatic compounds that avoids the halogenated precursor traditionally needed to make a boronic ester intermediate.
Together with major pharmaceutical firms, the Green Chemistry Institute established the Pharmaceutical Roundtable in 2005. It was formed as a working group made up of pharmaceutical industry representatives with a mission to identify and overcome common drug discovery and process chemistry challenges.
The group spent its first year singling out seven reactions and processes that need cost and environmental improvements, as well as a wish list of five reactions that chemists would like to add to their research arsenals (C&EN, July 24, 2006, page 36).
The roundtable issued a call for proposals last November to address the targeted reactions and processes and received an unexpected 32 international applications within a few weeks, says Pfizer's Peter J. Dunn, one of the roundtable's representatives. In February, the first grant of $130,000 was awarded to chemistry professor Jianliang Xiao of the University of Liverpool, in England, whose group is using the funds to develop a cleaner, more efficient method for hydrogenating amides to form amines (C&EN, Feb. 12, page 19).
"Because the response to the call for proposals was outstanding, we wanted to issue a second grant," Dunn says. By issuing the grants, the roundtable hopes "to continue encouraging mainstream chemists to address green chemistry problems," he adds. The group plans to issue a new call for proposals in August and anticipates awarding other grants for basic research in the next couple of years.
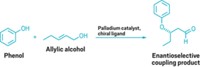
Maleczka and Smith will use their grant to advance their work on C???H activation of nonhalogenated arenes and heteroarenes using iridium catalysts and pinacolborane to form boronic ester intermediates. The intermediates cross-couple with aryl or alkenyl halides in subsequent palladium-catalyzed reactions to form unsymmetrical biaryl or vinyl arene compounds.
These one-pot activation/coupling reactions are of interest because they offer a direct synthesis of important pharmaceutical intermediates while eliminating the environmentally problematic halogenated aromatics commonly used to make the boronic ester intermediates.


Join the conversation
Contact the reporter
Submit a Letter to the Editor for publication
Engage with us on Twitter