Advertisement
Grab your lab coat. Let's get started
Welcome!
Welcome!
Create an account below to get 6 C&EN articles per month, receive newsletters and more - all free.
It seems this is your first time logging in online. Please enter the following information to continue.
As an ACS member you automatically get access to this site. All we need is few more details to create your reading experience.
Not you? Sign in with a different account.
Not you? Sign in with a different account.
ERROR 1
ERROR 1
ERROR 2
ERROR 2
ERROR 2
ERROR 2
ERROR 2
Password and Confirm password must match.
If you have an ACS member number, please enter it here so we can link this account to your membership. (optional)
ERROR 2
ACS values your privacy. By submitting your information, you are gaining access to C&EN and subscribing to our weekly newsletter. We use the information you provide to make your reading experience better, and we will never sell your data to third party members.
Materials
Bent DNAs Help Probe DNA-Protein Binding
New class of artificial DNAs could help assess structure-specific DNA recognition
by Celia Henry Arnaud
August 9, 2007
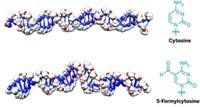
Many DNA-protein interactions involve structural distortions of the DNA helix. A new class of artificial DNAs, called "modular bent DNAs," could help systematically assess the role that DNA bending plays in these interactions, according to a recent study (J. Am. Chem. Soc., DOI: 10.1021/ja071335k).
Akira Matsuda, Satoshi Ichikawa, Shunpei Murata, and coworkers at Hokkaido University, Sapporo, Japan, create their bent DNA oligonucleotides by adding cyclic 2'-deoxyuridylate dimers connected by alkylene linkers. Oligonucleotides with shorter alkylene "clasps" have larger bending angles and are thermally less stable.
The researchers tested the bent DNAs by binding them to HMGB1, a protein known to interact with DNA-cisplatin adducts, which are themselves bent DNAs. The binding affinity of the bent DNA decreases as the linker length increases, and the oligonucleotide with a propylene bridge doesn't form a complex with the protein.
When an acyclic form of the dimer is used to bend the DNA, the bending angle is small and the oligonucleotide doesn't bind the protein, further suggesting the importance of DNA bending for protein binding.




Join the conversation
Contact the reporter
Submit a Letter to the Editor for publication
Engage with us on Twitter