Advertisement
Grab your lab coat. Let's get started
Welcome!
Welcome!
Create an account below to get 6 C&EN articles per month, receive newsletters and more - all free.
It seems this is your first time logging in online. Please enter the following information to continue.
As an ACS member you automatically get access to this site. All we need is few more details to create your reading experience.
Not you? Sign in with a different account.
Not you? Sign in with a different account.
ERROR 1
ERROR 1
ERROR 2
ERROR 2
ERROR 2
ERROR 2
ERROR 2
Password and Confirm password must match.
If you have an ACS member number, please enter it here so we can link this account to your membership. (optional)
ERROR 2
ACS values your privacy. By submitting your information, you are gaining access to C&EN and subscribing to our weekly newsletter. We use the information you provide to make your reading experience better, and we will never sell your data to third party members.
Synthesis
Aerobic, Copper-Templated Cross-Coupling
Unprecedented catalytic cycle offers a new pathway to C–C bond formation
by Stephen K. Ritter
December 12, 2007

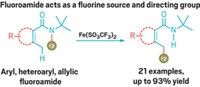
A highly selective synthesis of ketones that follows a novel carbon-carbon bond-forming reaction mechanism has been demonstrated by Janette M. Villalobos, Jiri Srogl, and Lanny S. Liebeskind of Emory University (J. Am. Chem. Soc., DOI: 10.1021/ja074931n). The synthesis, an offshoot of popular boronic acid-based cross-coupling reactions, could find utility in medicinal chemistry applications where complex substrates and products contain sensitive functional groups that must be handled under mild reaction conditions.
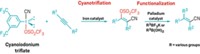
In 2000, Srogl and Liebeskind reported a unique coupling of thiol esters with boronic acids to produce ketones. That reaction, mediated by a palladium catalyst and a stoichiometric copper cocatalyst, operates at neutral pH and in the absence of oxygen. These conditions permit the use of substrates with functional groups that would not survive standard cross-coupling reactions, which normally need a base. All published thioorganic boronic acid cross-couplings to date require the palladium-copper combination and involve formation of a 1:1 intermediate complex of copper and the thiol ester.
Liebeskind's group has now determined that the same reaction can take place under aerobic conditions and without the palladium catalyst. The key is formation of the Cu(I)-thiol ester complex, which is oxidized by O2 in air to form a Cu(II/III) intermediate. This intermediate serves as a template for the thiol ester terminal group to couple with the boronic acid substituent group to form a ketone.
The residual copper thiolate complex next reacts with a second equivalent of boronic acid to form a thioether—the starting Cu(I) complex is regenerated in the process to complete the cycle. The researchers showed that a variety of thiol ester and boronic acid analogs are amenable to the coupling reaction, leading to 1:1 mixtures of ketones and thioethers in moderate yields.
"The combination of Cu(I) and O2 leading to a catalytic cycle involving Cu(II/III) is unusual in this kind of cross-coupling," says Carsten Bolm of RWTH Aachen University, in Germany, whose group is working on ligand-free copper-catalyzed cross-coupling reactions. "In conjunction with the template concept, I can well-imagine important synthetic applications."
Although the ketone is the more significant of the two products formed, both products can be tailored for specific applications, Liebeskind says. Because of the mild reaction conditions, "this chemistry could find use in a drug discovery setting where sensitive and complex structures are desired," he notes.
For example, the new chemistry could be used to react at specific biologically important thiol ester residues, such as acylated cysteines in proteins, Liebeskind says. Such reactions could provide a way to tag both the protein and the acyl residues in a site-specific fashion.
The Emory team continues to work on defining the potential scope of the reaction, Liebeskind adds. So far, this includes the synthesis of peptidyl ketones, in addition to the reported ketones and thioethers.


Join the conversation
Contact the reporter
Submit a Letter to the Editor for publication
Engage with us on Twitter