Advertisement
Grab your lab coat. Let's get started
Welcome!
Welcome!
Create an account below to get 6 C&EN articles per month, receive newsletters and more - all free.
It seems this is your first time logging in online. Please enter the following information to continue.
As an ACS member you automatically get access to this site. All we need is few more details to create your reading experience.
Not you? Sign in with a different account.
Not you? Sign in with a different account.
ERROR 1
ERROR 1
ERROR 2
ERROR 2
ERROR 2
ERROR 2
ERROR 2
Password and Confirm password must match.
If you have an ACS member number, please enter it here so we can link this account to your membership. (optional)
ERROR 2
ACS values your privacy. By submitting your information, you are gaining access to C&EN and subscribing to our weekly newsletter. We use the information you provide to make your reading experience better, and we will never sell your data to third party members.
Synthesis
Iron's Star Rising
For more sustainable chemical processing, chemists are exploring catalysts based on abundant and increasingly versatile iron
by Stephen K. Ritter
July 28, 2008
| A version of this story appeared in
Volume 86, Issue 30

PRECIOUS METALS currently are the basis of catalysts that are well understood by chemists and widely used in labs, industrial processes, and automobile catalytic converters. But the metals—such as platinum, palladium, and rhodium—are rare and expensive, and they can be toxic and difficult to recycle.
Catalysts enable some 90% of chemical processes, and chemists are always on the lookout for new ones. As they consider how to foster a more sustainable future, they are seeking alternatives that not only are greener and less expensive than current catalysts but also as effective or better.
Matthias Beller and coworkers Stephan Enthaler and Kathrin Junge at the Leibniz Institute for Catalysis at the University of Rostock, in Germany, make up one team of chemists that has been giving serious thought to possible alternatives. They have investigated the first-row transition metals iron, copper, zinc, and manganese as potential homogeneous catalysts that can contribute to a more sustainable chemical enterprise. In particular, they have latched onto iron as a "rising star" in the constellation of catalysts available to chemists.
"Iron offers significant advantages compared with precious metals," Beller says. It is the second most abundant metal in Earth's crust after aluminum, he notes. That means it's much less expensive than other metals.
Iron also permits the use of nitrogen ligands instead of phosphines, which are generally needed for precious-metal catalysts, he says. Another benefit is that iron is less toxic to people and the environment than precious metals. And thanks to how easily it changes oxidation state and its Lewis acid character, iron is already a proven effective catalyst in a few industrial applications, he adds.
In a recent study, Beller, Enthaler, and Junge compared the market prices of common catalyst metals platinum, palladium, rhodium, iridium, and ruthenium with the price of iron (Angew. Chem. Int. Ed. 2008, 47, 3317). The prices of the precious metals at the time of the study ranged from $1,365 per mol for ruthenium to $28,172 per mol for rhodium. The price for iron was 0.9 cents per mol.
The comparison is made between pure metals and scrap iron and does not take into account the cost and potential toxicity of ligand systems, which can be quite expensive, particularly for chiral phosphine ligands. But the point Beller and coworkers make is striking: If chemists can devise iron catalysts that match the reactivity, selectivity, and substrate scope of precious-metal catalysts—which hasn't been generally achieved before now—significant cost and environmental savings await.
AS PART of their study, Beller and coworkers examined recent developments in iron-catalyzed organic synthesis. One example they point to is the research of Carsten Bolm of Rhenish-Westphalian Technical University, in Aachen, Germany, whose group is working on iron-catalyzed cross-coupling reactions and iron-catalyzed oxidations of saturated hydrocarbons and sulfides. Bolm's group has written two reviews of iron catalysis (Chem. Rev. 2004, 104, 6217; Chem. Soc. Rev. 2008, 37, 1108).
In one reaction, Bolm and Arkaitz Correa paired FeCl3 with an ethylenediamine ligand to convert aryl halides and pyrazole, indole, or other nitrogen-containing substrates into N-aryl compounds. The products contain important structural motifs found in natural products and bioactive synthetic compounds, and they are essential intermediates in preparing pharmaceuticals and other fine chemicals, Bolm notes. The method offers an alternative to palladium- and copper-catalyzed C–N coupling reactions, he adds. Although copper is a first-row transition metal, it is in high demand and its cost has shot up lately, making it relatively much more expensive than iron.
"Our research has established a useful starting point for investigating future applications of iron-catalyzed arylation reactions," Bolm says. In fact, his group has already expanded the scope of the reactions to synthesize O- and S-aryl products. And in the lab's most recent development, Bolm, Correa, and coworker Mónica Carril used their iron catalyst in a preliminary study of a Sonogashira reaction to couple phenylacetylene with phenyl iodide to form diphenylacetylene and related compounds (Angew. Chem. Int. Ed. 2008, 47, 4862). The Sonogashira reaction currently is the most popular method for making sp2-sp carbon-carbon bonds (=C–C≡C–), the researchers point out.
"The numerous advantages of iron make it highly attractive as a catalyst metal," Bolm says. "Given more time, iron catalysis will surely become an even more powerful tool for organic synthesis and find its place in many more industrial applications."
In another example of iron's capabilities, Beller and coworkers cite work by Cornell University's Paul J. Chirik, who uses iron complexes to mediate the hydrogenation and hydrosilylation of alkenes and alkynes. Chirik's group has developed an iron catalyst with a bis(imino)pyridine ligand that hydrogenates olefins such as 1-hexene at rates comparable with those of standard palladium, rhodium, and iridium catalysts (Organometallics 2008, 27, 1470).
"The area of iron catalysis is undergoing explosive growth," Chirik says. "It is not only interesting from a cost and environmental standpoint, but also from a fundamental perspective." The ability to control one-electron chemical processes with iron catalysts is a real challenge and requires different ways of thinking about ligand design, he notes. "We've had a lot of fun wrestling with these new problems and challenges," he says.
But iron catalysis is still in its infancy, Chirik adds. "As the prices for precious metals continue to rise, the bar lowers for an effective iron catalyst, especially considering that catalyst recycling costs energy and time, generates waste, and is almost never 100% effective," he says. "I think if you can get in the ballpark with iron, a lot of people will consider using it or another first-row metal before a precious metal."
Beller's analysis and the examples he and his coworkers explore focus on homogeneous catalysts, which enable solution-phase organic transformations such as those used in medicinal chemistry and in pharmaceutical and fine chemicals process chemistry. But iron catalysts are applicable to heterogeneous reactions too.
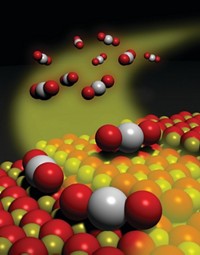
FOR EXAMPLE, in a study involving heterogeneous iron catalysts, Jens K. Nørskov of the Technical University of Denmark and colleagues identified alternatives to metal particles such as palladium-silver alloys that are used in hydrogenation reactions (Science 2008, 320, 1320).
The team used computational methods, catalyst-screening protocols, and reaction chemistry to evaluate combinations of metals as potential catalysts for hydrogenation of acetylene to ethylene. This important industrial process, which typically uses PdAg catalysts, selectively converts trace amounts of acetylene in ethylene streams before ethylene is polymerized.
The study led to several alternatives—including NiZn, FeZn, NiZn3, and FeZn3 alloys—with selectivity and reactivity profiles similar to or better than those of PdAg catalysts. But the nickel and iron catalysts potentially cost thousands of dollars per kilogram less than the PdAg catalysts, the researchers point out, and they contain metals with relatively low toxicity.
Nørskov says he "shares the ambition" of Beller and others of finding non-precious-metal alternatives. "The special angle in our story," he notes, "is that we use electronic structure calculations to identify promising heterogeneous catalyst candidates in a procedure that also provides insight into the important parameters characterizing a good surface catalyst."
ANOTHER APPROACH, to developing better iron catalysts has been to mimic iron-catalyzed biological processes. Iron is a key metal in a number of metalloproteins that transport or metabolize small molecules such as oxygen, nitrogen, and methane. Chemists are starting to take advantage of the insight gained by studying nature's approach in these processes and are refining their ability to create metal-ligand systems that can match the catalytic activity of enzymes.
For example, iron plays a key role in cytochrome P450 enzymes that are found in all types of life forms and in methane monooxygenase (MMO) bacterial enzymes. In humans, cytochrome P450 is involved in making cholesterol, steroids, and other lipids, and it also metabolizes drugs into oxidized forms that can be excreted from the body. MMO converts methane to methanol, which certain bacteria use to live on.
Both cytochrome P450 and MMO need oxygen to carry out hydroxylation reactions that activate and convert C–H bonds to C–OH bonds. The two enzymes have structural differences at their iron cores: Cytochrome P450 has a reaction center made up of a porphyrin ring with an iron ion in the middle, a group known as heme; the soluble form of MMO has a reaction center in which two iron atoms are bridged by two oxygen atoms and the metal atom coordination is completed by histidine and carboxylate amino acid ligands—a nonheme structure.
It's difficult to develop simple artificial versions of enzymes, because the whole enzyme, not just the metal active site, is usually required to carry out a chemical conversion. For example, chemists have learned by modeling cytochrome P450 that the porphyrin ring doesn't offer the same protective surroundings as the enzyme, and the lack of that protection leads to degradation of potential catalysts. But models based on MMO and related nonheme iron enzymes are proving to be more successful.
Beller and coworkers point to the research of Lawrence Que Jr. at the University of Minnesota, Twin Cities, who is using bioinspired ligands to make iron oxidation catalysts. A decade ago, Que's group was the first to develop a synthetic model of a nonheme iron active site—a single iron atom with a tris(2-pyridylmethyl)amine ligand—capable of catalyzing the stereospecific hydroxylation of alkanes using hydrogen peroxide as an oxidant. Que and coworkers subsequently reported the first crystal structure of a mononuclear Fe=O complex, which serves as a model for the active form of many nonheme iron enzymes.
In a continuation of the work, Que and Eckard Münck of Carnegie Mellon University and coworkers recently reported a new iron complex with an Fe2O2 core and nitrogen-based ligands that provides one of the best models for studying MMO's mechanism of action (C&EN, Dec. 24, 2007, page 35). The researchers have concluded that the Fe2O2 core opens up to form a terminal Fe=O unit that reacts with CH4. In a synthesis based on those studies, Que's group has shown that monoiron complexes with biomimetic nitrogen-based ligands can use H2O2 to selectively oxidize olefins into diols rather than form epoxides, which are the typical products of such oxidations.
In the same year that Que's model was announced, Yuzo Nishida and coworkers at Japan's Yamagata University reported a second example of a bioinspired nonheme iron catalyst capable of hydroxylating alkanes. Nishida's version uses a different ligand, N,N′-dimethyl-N,N′-bis(2-pyridiylmethyl)ethane-1,2-diamine, or "mep" for short.
In a novel twist, M. Christina White and coworkers at the University of Illinois, Urbana-Champaign, last year incorporated pyrrolidine rings into mep's backbone to make it more rigid (C&EN, Nov. 5, 2007, page 8). In contrast to the simpler nonheme catalysts developed by Que and Nishida, the more rigid catalyst can perform highly selective oxidations in preparative yields, and it can do so without the need for directing, activating, or protecting groups.
White's iron catalyst has created quite a stir in the organic chemistry community for being the first catalyst system for selective and predictable oxidation of unactivated aliphatic C–H bonds at tertiary carbon centers in complex molecules. Specifically, White and graduate student Mark S. Chen oxidized the antimalarial natural product (+)-artemisinin at only one of the compound's five tertiary C–H bonds, forming (+)-10β-hydroxyartemisinin. The 54% yield they obtained compares favorably with the 47% yield of a microbial synthesis that takes four days. The synthesis may be the best example so far of the potential of iron catalysts to replace precious-metal catalysts in medicinal chemistry.
"The recent surge in the number of iron catalysts that display hydrogenation and cross-coupling reactivity, which is traditionally reserved for precious metals such as palladium and rhenium, is tremendously exciting," White says. "Many opportunities exist in this area for new reaction discovery and mechanistic understanding."

White and Chen's "elegant demonstration" of the ability to carry out highly selective, nonheme iron-catalyzed oxidations is "a very exciting advancement in organic synthesis," comments Steven J. Brickner, a research fellow in antibacterials chemistry at Pfizer. "The exquisite selectivity seen in their hydroxylations is exceedingly rare outside the realm of enzymatic reactions," he says. White has formed a research collaboration with Pfizer to further explore the scope of the reaction and to optimize the chemistry.
Advertisement
Carl P. Decicco, vice president of discovery chemistry at Bristol-Myers Squibb, agrees with Brickner's assessment. "What we are trying to do in drug discovery and process development is to be as efficient as possible—both cost-efficient and time-efficient—and in the modern world to be environmentally friendly," Decicco notes. "These things are on the minds of everyone doing organic synthesis."
White's catalyst has gained a lot of attention because it reaches those goals, he says. "Not only is it efficient, but perhaps for the first time ever it remarkably shows how to take a substrate that has very little in the way of activated atoms and be able to selectively oxidize them. It's an excellent discovery."

Despite the advances being made in iron catalysis, the only well-known example of an iron-catalyzed industrial reaction is the Haber-Bosch nitrogen-fixation process to make ammonia and other nitrogen-containing compounds. Haber-Bosch chemistry is complex, requiring multiple steps and several catalysts to first generate hydrogen from natural gas or methane. The final step involves passing the hydrogen and nitrogen over a heterogeneous iron oxide catalyst—an activated form of the mineral magnetite—at high temperature and high pressure to make ammonia. Iron oxide is also used in oil refinery processes such as the water-gas shift reaction and Fischer-Tropsch synthesis.
"The other known catalytic reactions with iron are either limited in scope or do not qualify for practical applications," Leibniz Institute's Beller says. "In this respect, the use of iron as a catalyst is underdeveloped." That makes using a catalyst as simple as iron oxide—essentially rust—very enticing because it could become the basis of a whole new thrust in chemistry, Beller notes.
Many chemical processes could benefit from iron catalysis in the coming years. In the area of transportation fuels, new catalysts will be needed to improve desulfurization and denitrogenation of gasoline and diesel fuel to reduce automobile pollution as lower grades of crude oil are increasingly used. Effective catalysts for C–H activation of simple alkanes to make longer chain hydrocarbons could be crucial in processes that convert plant sugars into biofuels. And as White's work shows, C–H activation is already a hot area of research aimed at the synthesis of complex molecules for pharmaceuticals and fine chemicals.
Still another target is developing iron nanoparticles that can catalyze chemical syntheses and the remediation of contaminated soil and groundwater. And even for the well-established Haber-Bosch reaction, switching from a heterogeneous iron catalyst to a soluble homogeneous catalyst might permit the reaction to operate at lower temperature and pressure and significantly reduce energy costs.
Iron complexes are becoming a much more interesting and viable choice for catalysis, says Beller, whose own work includes iron-catalyzed reductions of carbonyl groups to make alcohols under mild conditions. "Clearly, the reported iron catalyst systems are still far from immediate industrial applications," he notes. And the chemical industry will not shift away from precious-metal catalysts to iron or other alternatives until it is advantageous to do so. "But for the midterm future, we can expect a significant increase in the use of iron catalysis in organic synthesis," Beller concludes.



Join the conversation
Contact the reporter
Submit a Letter to the Editor for publication
Engage with us on Twitter