Advertisement
Grab your lab coat. Let's get started
Welcome!
Welcome!
Create an account below to get 6 C&EN articles per month, receive newsletters and more - all free.
It seems this is your first time logging in online. Please enter the following information to continue.
As an ACS member you automatically get access to this site. All we need is few more details to create your reading experience.
Not you? Sign in with a different account.
Not you? Sign in with a different account.
ERROR 1
ERROR 1
ERROR 2
ERROR 2
ERROR 2
ERROR 2
ERROR 2
Password and Confirm password must match.
If you have an ACS member number, please enter it here so we can link this account to your membership. (optional)
ERROR 2
ACS values your privacy. By submitting your information, you are gaining access to C&EN and subscribing to our weekly newsletter. We use the information you provide to make your reading experience better, and we will never sell your data to third party members.
Environment
Side Effects
Pharmaceuticals have been finding their way into our environment for a long time, but just what are they doing there?
by Bethany Halford
February 25, 2008
| A version of this story appeared in
Volume 86, Issue 8

NO ONE EVER planned for fish to take birth control pills. But they are. As treated wastewater flows into rivers and streams every day, fish all over the world get a tiny dose of 17α-ethinylestradiol, a synthetic steroidal estrogen that's used in birth control pills. They also get a little sip of the anticonvulsant carbamazepine, a nip of the antidepressant fluoxetine, and a taste of hundreds of other drugs that we take to make our lives better.
Every drug begins its life as a promise—a promise to fight disease or improve our quality of life. It wends its way through the discovery process and clinical trials until it ends up in our bodies, ready to do its job.
But that's not the end of the story. A drug doesn't simply disappear once it has served its purpose. People and animals excrete pharmaceuticals and their metabolites, which then find their way into the environment through a variety of routes—treated wastewater, agricultural runoff, and biosolids and manure that are used as fertilizers. Pharmaceuticals also enter the environment when people dispose of medications by flushing them down the toilet or pouring them down the drain.
Concentration-wise, pharmaceuticals represent just a small fraction of the thousands of man-made chemicals in the environment, including everything from pesticides to personal care products. Even though medications have been in the environment for as long as people have been taking them, it's only through the advent of advanced analytical instrumentation and techniques that scientists have been able to detect them in the wild.
The small concentrations, however, belie the potentially powerful effects of these compounds. Pharmaceuticals are specifically designed to elicit a biological response at very low levels, and scientists are increasingly becoming aware of how medications let loose on the environment may have effects no one ever anticipated.
By far, the most dramatic example of this kind of pharmaceutical pollution has been the effect of estrogenic compounds on fish. In the 1990s, scientists working in the U.K. noted that male fish living downstream from wastewater treatment plants were becoming feminized. They were making proteins associated with egg production in female fish, and they were developing early-stage eggs in their testes. Feminized male fish have now been observed in rivers and streams in the U.S. and Europe.
Research led by John Sumpter, an ecotoxicologist at England's Brunel University, linked the feminizing phenomenon to the presence of estrogenic compounds, such as the synthetic birth control compound 17α-ethinylestradiol and the natural hormone 17β-estradiol, in the water (Environ. Sci. Technol. 1998, 32, 1549). Municipal wastewater treatment plants don't completely break down these estrogenic compounds or their metabolites. For example, 17α-ethinylestradiol is excreted in the form of glucuronide and sulfate conjugates. Bacteria in the wastewater treatment process cleave these conjugate groups, regenerating the original estrogenic compound.
"It doesn't take a lot of estrogen to feminize male fish," says Karen A. Kidd, a biology professor at the Canadian Rivers Institute, University of New Brunswick. "If you can measure the estrogen in the water, then that's enough to cause an effect, and we can measure it at very low parts-per-trillion concentrations."
Kidd recently spearheaded a research project to study the overall impact of estrogenic compounds on fish (Proc. Natl. Acad. Sci. USA 2007, 104, 8897). "No one knew what feminization meant for the fish population," Kidd explains. "Can feminized males still successfully reproduce, or is there going to be an impact on the number of fish in the rivers? That's the big question we set out to address."
FOR THREE SUMMERS, Kidd and her colleagues spiked a lake in Canada's Experimental Lakes Area with 17α-ethinylestradiol at a concentration of 5 ppt—a concentration that has been measured in municipal wastewaters and in river waters downstream of discharges. During the autumn that followed the first addition of the estrogenic compound, the researchers observed delayed sperm cell development in male fathead minnows—the freshwater equivalent of a canary in a coal mine. A year later, the male fathead minnows were producing eggs and had largely stopped reproducing. The minnow population began to plummet. The decline continued for an additional three years until the fish had all but disappeared from the lake.
The fathead minnow wasn't the only fish to feel the effects of the trace amounts of birth control. The population of lake trout, which feed on smaller fish, fell by about 30%. "The numbers of lake trout dropped not because of direct exposure to the estrogens but because they lost their food supply," Kidd says.
But Kidd's story is not all doom and gloom. In 2006, three years after her team stopped adding 17α-ethinylestradiol to the lake, the fathead minnow population rebounded. "So given enough time, once you remove the estrogens from a system, the fish can recover to their original population size," Kidd notes.
Although estrogenic compounds may have the most dramatic effect on fish, they aren't the only pharmaceuticals that have been implicated in the pollution of aquatic environments. Laboratory studies have shown that the antidepressant fluoxetine, or Prozac, can slow the development of fish and frogs. The anticonvulsant carbamazepine affects the emergence of mosquito-like insects that are a popular food source for certain fish.
"The challenge is that we don't have a good understanding of" a lot of drugs that are being used, Kidd says. "We've looked at only a handful of the drugs that are actually used and discharged into our waters. There are just a lot of unknowns in this field right now."
And it's not just aquatic organisms that are feeling the effects of pharmaceutical pollution. One study traced massive die-offs of vultures in Asia to the veterinary use of diclofenac, a nonsteroidal, anti-inflammatory (Nature 2004, 427, 630).
Diclofenac is frequently used to treat domestic livestock in India and Pakistan. When these animals die from disease or injury, they're typically left for scavengers. An international team of scientists, led by J. Lindsay Oaks of Washington State University, found that the vultures were consuming these carcasses and dying from renal failure and visceral gout caused by diclofenac poisoning.
Scientists also worry that the massive amounts of antibiotics used to treat livestock may be creating antibiotic-resistant microbes. "There's a whole other source of pharmaceutical pollution that really needs attention, and that's livestock use, which generates an estimated 500 million tons of waste each year," says Dana W. Kolpin, a research hydrologist at U.S. Geological Survey (USGS) who studies emerging contaminants in the environment.
Kolpin points out that livestock manure is full of antibiotics, synthetic and biogenic hormones, and other veterinary medicines. Farmers use sludge generated by sewage treatment plants as a fertilizer and a source of nutrients for crops, but this material also contains excreted medications.
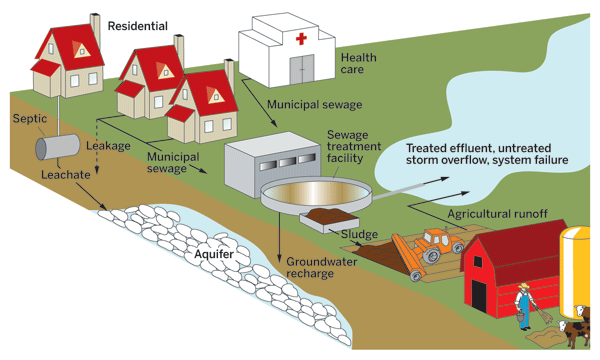
Pharmaceuticals and their metabolites enter municipal sewage systems and aquifers from homes, health care facilities, and farms.
SCIENTISTS KNOW that these pharmaceuticals can travel into the environment as agricultural runoff and soil contaminants. A new report from chemistry professor Chad A. Kinney of Colorado State University, Pueblo, shows that earthworms living in the contaminated soil can take up some of these pharmaceuticals, notably the antibiotic trimethoprim, albeit in small amounts (Environ. Sci. Technol., DOI: 10.1021/es702304c). "It shows there is uptake into earthworms that leads to a potential pathway up the food chain, as earthworms are a major food source for many higher organisms," says Kolpin, who was a coauthor of the report. Studies have also shown that pharmaceuticals can be taken up into crop plants (J. Agric. Food Chem. 2006, 54, 2288).
Researchers continue to suss out the subtle effects of pharmaceuticals on the environment, such as antibiotic resistance and changes in feeding and mating behaviors. But getting a handle on the whole problem is a huge task. Not only do researchers need to consider the parent compounds in their analyses, but they also have to look at metabolites and at the transformation products that are formed either during treatment or from natural processes taking place in the environment, such as microbial degradation and photolysis.
"If you just look at the parent compound, that's only giving you a piece of the story," Kolpin explains. "Each compound can break down and form new environmental contaminants, which can become much more mobile and much more persistent."
"Sometimes there are cases for which the biodegradation product is more toxic than the parent compound," adds Diana Aga, an analytical chemist at State University of New York, Buffalo, who is developing tools to detect trace levels of pharmaceuticals.
Scientists also have to keep in mind that pharmaceuticals aren't isolated in the environment, Kolpin notes. They could be acting in concert with a surfactant, another pharmaceutical, or some other environmental contaminant.
The Food & Drug Administration requires pharmaceuticals to undergo an environmental risk assessment before they can go on the market. These tests are performed on both terrestrial and aquatic organisms, but they're usually short-term tests that measure how much of a compound is required to kill an organism outright or stunt its growth within a matter of days.
"If we're going to identify the compounds that are going to be problematic, we need to be looking for the right things," says Bryan Brooks, an environmental science professor at Baylor University. For example, he explains, it takes a lot of 17α-ethinylestradiol to kill an aquatic organism, so by current testing standards, the compound would appear to have a very low potential risk. But feminization of male fish—something those short-term tests would have never detected—occurs at very low concentrations of the drug.
THE BIG QUESTION is whether pharmaceutical pollution has any impact on human health. While trace amounts of pharmaceuticals do find their way into drinking water, studies indicate that the concentrations are far too small to elicit any appreciable effect.
Fetal exposure to certain pharmaceuticals is also a cause for concern. An extreme example would be exposure to thalidomide or to a chemotherapeutic drug, says Christian G. Daughton, chief of the environmental chemistry branch at the Environmental Protection Agency's National Exposure Research Laboratory. He adds, however, that "the doses that a fetus would get from its mother ingesting several liters of water a day are still orders of magnitude below the dosages that are known to cause effects."
Daughton says that toxicologists are now trying to understand the effects of continual sustained exposure to multiple chemicals, each present at a very low level. "It could be that the chemical stress that's put on any organism is the result of minute stresses of a multitude of chemicals," whether they're synthetic or naturally occurring compounds, he says.
Francesco Pomati, a toxicologist at Australia's University of New South Wales, and colleagues at Italy's University of Insubria recently discovered that a low-concentration mixture of 13 drugs—including a chemotherapeutic agent and several antibiotics—can inhibit the growth of human embryonic kidney cells in vitro (Environ. Sci. Technol. 2006, 40, 2442). It's important to note, however, that the concentrations used were those found in the environment, not in drinking water.
"Studies conducted to date suggest that it is highly unlikely that the quantities of pharmaceuticals detected in the environment would be harmful to human health," says Ken Johnson, senior vice president with Pharmaceutical Research & Manufacturers of America.
"I think it's unambiguous that the trace pharmaceuticals we're seeing in drinking water have no human health problems so far," adds Shane Snyder, an environmental toxicologist at the Southern Nevada Water Authority. A few months ago, Snyder published an analysis of water from 20 drinking water utilities across the U.S. These utilities were treating water known to contain wastewater that had come from a sewage treatment plant upstream.
Notably, Snyder's team did not detect any of the estrogenic compounds that have been implicated in the feminization of fish in either the source water or the treated drinking water. They did find that drinking water from at least half of the treatment facilities contained ibuprofen, carbamazepine, the antiepileptic dilantin, and the antianxiety drug meprobamate, but each occurred in extremely low concentrations—in the parts-per-trillion range.
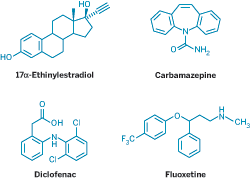
Medications that scientists think may have negative effects on the environment.
"The treatment processes we have are highly effective," Snyder concludes. He points out that we're seeing more pharmaceuticals in our environment because we're getting better at detecting them, not necessarily because there are more of them. It's therefore important, he says, to develop toxicologically based limits for pharmaceuticals in our water. "If we ignore concentration and say presence or absence is our litmus test, then there will be no end to that," Snyder says. "Detection does not infer health risk and nondetection does not ensure safety."
Advertisement
Snyder's study also assessed the removal of pharmaceuticals from drinking water, using both conventional and advanced drinking water treatment processes. Conventional treatments, such as coagulation, flocculation, and filtration, were basically ineffective. Chlorination proved to be better, removing about half of the compounds considered in the study. Advanced treatments, such as ozonation, activated carbon, and reverse osmosis and nanofiltration membranes, worked well, but these methods are expensive, and they're generally used to treat drinking water, not wastewater.
"YOU HAVE TO keep in mind that sewage treatment plants were originally designed a long time ago to improve the aesthetic quality of treated sewage and to reduce the incidence of disease—to reduce odor and make the water look better and get rid of bacteria and viruses," EPA's Daughton says. "They were never engineered to remove synthetic substances."
Whether or not sewage treatment plants should be equipped to remove pharmaceuticals is a matter of some controversy. "There's this huge risk-benefit equation that's very difficult to address," Daughton explains.
"I think for a lot of drugs out there, we are probably more concerned than we should be," says Alistair Boxall, an environmental chemist at the University of York, in England. "I think there are examples of substances that perhaps we do need to look at a bit further, but I'm not convinced we should be putting advanced treatment mechanisms on every sewage treatment plant just to get rid of pharmaceuticals."
Snyder agrees. "I would be very cautious about building energy-intensive wastewater treatment plants," he tells C&EN.
"With regard to pharmaceuticals in the environment, I do believe that we have the treatment technologies available to address these problems," says Nancy G. Love, a professor of civil and environmental engineering at the University of Michigan . "The challenge comes in improving the engineering implementation of the technologies" to make them more cost-effective, she adds.
Even though pharmaceutical pollution is a problem we are equipped to deal with, the solution may not be so simple if the contaminants are causing antibiotic resistance, Love says. "We do not necessarily have the technology to design antibiotics that are not vulnerable to generating antibiotic-resistant characteristics in microbes. And if pollution is an inducer of antibiotic resistance, then we have a larger problem on our hands than we realize."
Scientists working in this area agree that more research needs to be done on all aspects of pharmaceuticals' effects in the environment. "We need to determine which compounds or sets of compounds are the worst players, and then we need to make the decision whether these things need to be removed before they get into the environment," USGS's Kolpin says. "There's more information needed before any sort of policy or regulatory decision can be made. Otherwise, I'm afraid it will be ineffective or unnecessary."




Join the conversation
Contact the reporter
Submit a Letter to the Editor for publication
Engage with us on Twitter