Advertisement
Grab your lab coat. Let's get started
Welcome!
Welcome!
Create an account below to get 6 C&EN articles per month, receive newsletters and more - all free.
It seems this is your first time logging in online. Please enter the following information to continue.
As an ACS member you automatically get access to this site. All we need is few more details to create your reading experience.
Not you? Sign in with a different account.
Not you? Sign in with a different account.
ERROR 1
ERROR 1
ERROR 2
ERROR 2
ERROR 2
ERROR 2
ERROR 2
Password and Confirm password must match.
If you have an ACS member number, please enter it here so we can link this account to your membership. (optional)
ERROR 2
ACS values your privacy. By submitting your information, you are gaining access to C&EN and subscribing to our weekly newsletter. We use the information you provide to make your reading experience better, and we will never sell your data to third party members.
Materials
Two-For-One Catalyst
Immobilizing a transition-metal complex in silver yields synergistic properties
by Stephen K. Ritter
August 28, 2008

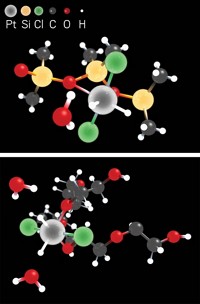
By entrapping a soluble rhodium complex inside a matrix of silver atoms, a team of Israeli chemists has initiated a new approach to heterogeneous catalysis (J. Am. Chem. Soc., DOI: 10.1021/ja804112z). Chemists often harness soluble homogeneous catalysts for surface-based heterogeneous reactions by fixing the catalyst on an organic polymer or an inorganic oxide such as silica, which are insulating materials. The catalyst-in-metal composite developed by Itzik Yosef, Raed Abu-Reziq, and David Avnir at Hebrew University of Jerusalem marks the first time a homogeneous catalyst has been "heterogenized" in a conducting material.

Avnir's group has previously developed "organically doped metals" by using solution processing to trap small organic molecules or polymers inside a matrix of metal atoms. Immersing the organic molecules in the conducting metal framework's "sea of electrons" significantly alters the properties of both components, Avnir says.
Trapping a catalyst in a metal takes the concept a step further. Entrapment of the rhodium complex enhances its catalytic activity, presumably by altering its electron donor-acceptor properties and modifying substrate binding.
This catalyst entrapment process "is a watershed development in science," comments organometallic chemist Howard Alper of the University of Ottawa, whose research includes rhodium-complexed dendrimers as heterogeneous catalysts. "The different properties of the entrapment entity open the way to new catalytic processes, as well as the improvement of existing ones," Alper says.
To make the material, the researchers added zinc powder to a solution containing a rhodium phosphine cyclooctadiene catalyst and silver nitrate. They believe that the silver cations attract negatively charged sulfonate groups on the phosphine ligand. The zinc reduces the silver cations, and as silver crystallites form they aggregate and precipitate out of the solution, taking the catalyst with it. The rhodium complex ends up entrapped within the three-dimensional metal matrix, as opposed to being adsorbed on the metal surface.
The nanoporous nature of the composite allows reactants to flow in and products to flow out, similar to what happens with porous catalyst materials such as zeolites and metal-organic frameworks. The rhodium catalyst remains trapped and doesn't leach out, which the team confirmed by solubility studies and electron microscopy.
The researchers tested the catalyst by hydrogenating styrene to ethylbenzene and diphenylacetylene to stilbene. The composite catalyst outperformed the rhodium catalyst alone and the rhodium catalyst adsorbed on silver; pure silver, used as a control, showed no reactivity.
"The unique synergism of the catalyst-in-metal system affords a new way to tailor catalyst properties," says materials scientist Bakul C. Dave of Southern Illinois University, Carbondale. The composite has "significant potential for commercial applications as a novel catalyst with enhanced activity," he adds.
It should be possible to entrap any type of catalyst in silver or other metals by the technique, Avnir says. For example, the Israeli group plans to explore trapping an oxidation catalyst in silver, which is itself an oxidation catalyst. The composite material could lead to a "killer" commercial oxidation system, Avnir believes.
Another potential application for the material is in carrying out multiple reaction steps in one vessel, Avnir notes. For example, a transition-metal catalyst could mediate one transformation followed by silver catalyzing a subsequent reaction, or vice versa.




Join the conversation
Contact the reporter
Submit a Letter to the Editor for publication
Engage with us on Twitter