Advertisement
Grab your lab coat. Let's get started
Welcome!
Welcome!
Create an account below to get 6 C&EN articles per month, receive newsletters and more - all free.
It seems this is your first time logging in online. Please enter the following information to continue.
As an ACS member you automatically get access to this site. All we need is few more details to create your reading experience.
Not you? Sign in with a different account.
Not you? Sign in with a different account.
ERROR 1
ERROR 1
ERROR 2
ERROR 2
ERROR 2
ERROR 2
ERROR 2
Password and Confirm password must match.
If you have an ACS member number, please enter it here so we can link this account to your membership. (optional)
ERROR 2
ACS values your privacy. By submitting your information, you are gaining access to C&EN and subscribing to our weekly newsletter. We use the information you provide to make your reading experience better, and we will never sell your data to third party members.
Materials
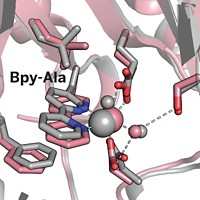
Unnatural Amino Acid Transforms Protein
Amino acid switch turns DNA-binding protein into DNA-cleaving protein
by Celia Henry Arnaud
September 24, 2008
By replacing a single naturally occurring amino acid with an unnatural amino acid, chemists at Scripps Research Institute have turned a protein that only binds to DNA into one that also cleaves the DNA specifically at the site next to the unnatural amino acid. Changing the location of the unnatural amino acid causes DNA cleavage at different spots. The technique could be used to map interactions between proteins and their DNA partners.
Chemistry professor Peter G. Schultz and graduate student Hyun Soo Lee perform this transformation on Escherichia coli catabolite activator protein (CAP), a homodimer that binds to a 22-base-pair site. They insert bipyridinyl alanine (Bpy-Ala) in place of a lysine that lies at the protein-DNA interface near both ends of the binding site in the dimeric protein (J. Am. Chem. Soc., DOI: 10.1021/ja804653f). The bipyridinyl group chelates transition-metal ions, which Schultz and Lee expected would induce oxidative cleavage of the DNA backbone next to the unnatural amino acid.
When the researchers incubate the modified protein with metal ions, a reducing agent, and DNA containing the binding sequence, they do indeed see cleavage of the DNA backbone in the expected locations.
The researchers suggest that similar methods could be used to convert any DNA-binding protein into a DNA-cleaving protein.




Join the conversation
Contact the reporter
Submit a Letter to the Editor for publication
Engage with us on Twitter