Advertisement
Grab your lab coat. Let's get started
Welcome!
Welcome!
Create an account below to get 6 C&EN articles per month, receive newsletters and more - all free.
It seems this is your first time logging in online. Please enter the following information to continue.
As an ACS member you automatically get access to this site. All we need is few more details to create your reading experience.
Not you? Sign in with a different account.
Not you? Sign in with a different account.
ERROR 1
ERROR 1
ERROR 2
ERROR 2
ERROR 2
ERROR 2
ERROR 2
Password and Confirm password must match.
If you have an ACS member number, please enter it here so we can link this account to your membership. (optional)
ERROR 2
ACS values your privacy. By submitting your information, you are gaining access to C&EN and subscribing to our weekly newsletter. We use the information you provide to make your reading experience better, and we will never sell your data to third party members.
Synthesis
Chemical Beaver Tale
Researchers build an enantiomerically pure component of the beaver's scent gland
by Sarah Everts
February 25, 2009

The Canadian beaver's scent glands were once a source of pricy perfumes, and extracts from the glands continue to be used in homeopathic medicine for treating hysteria. But on a different note, researchers in Germany have synthesized a component of the odoriferous cocktail through a methodology that uses sugar groups to confer chirality in organic molecules (Angew. Chem. Int. Ed., 10.1002/anie.200805606).
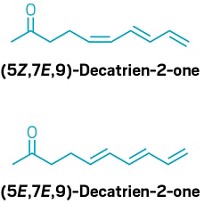
Horst Kunz and colleagues at the University of Mainz report the first enantioselective synthesis of an all-cis nupharamine found in the beaver's scent gland. In one of the 10 synthesis steps, a bulky pivaloyl-decorated sugar is attached to an intermediate enolate (shown) that eventually leads to an indole moiety. The presence of the bulky carbohydrate directs cis protonation of the enolate; without the sugar, trans protonation occurs.
Although the team hoped a tantalizing aroma would emerge from their products, the trans epimer smelled like stale fish and the cis epimer was odorless, Kunz says. About 40 compounds are in the gland extract, he adds, but the distinctive scent hasn't been reconstructed using the synthetic components. The long-sought-after odor is "still a secret of the beaver," Kunz notes.



Join the conversation
Contact the reporter
Submit a Letter to the Editor for publication
Engage with us on Twitter