Advertisement
Grab your lab coat. Let's get started
Welcome!
Welcome!
Create an account below to get 6 C&EN articles per month, receive newsletters and more - all free.
It seems this is your first time logging in online. Please enter the following information to continue.
As an ACS member you automatically get access to this site. All we need is few more details to create your reading experience.
Not you? Sign in with a different account.
Not you? Sign in with a different account.
ERROR 1
ERROR 1
ERROR 2
ERROR 2
ERROR 2
ERROR 2
ERROR 2
Password and Confirm password must match.
If you have an ACS member number, please enter it here so we can link this account to your membership. (optional)
ERROR 2
ACS values your privacy. By submitting your information, you are gaining access to C&EN and subscribing to our weekly newsletter. We use the information you provide to make your reading experience better, and we will never sell your data to third party members.
Environment
Creating Fuel From Water
Sustainable Energy: New materials aim to improve the path to dihydrogen
by Jyllian N. Kemsley
May 3, 2010
| A version of this story appeared in
Volume 88, Issue 18

Two research groups report new materials aimed at achieving a long-standing research goal: the ability to use fuel cells to generate H2, a clean-burning sustainable fuel, from water.
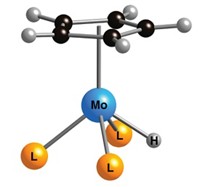
Traditional catalysts for H2 generation are based on platinum, but the metal is expensive, globally limited in supply, and easily poisoned. Working to find a less expensive alternative, graduate student Hemamala I. Karunadasa and chemistry professors Christopher J. Chang and Jeffrey R. Long of the University of California, Berkeley, have developed a molybdenum oxo catalyst containing the ligand 2,6-bis[1,1-bis(2-pyridyl)ethyl]pyridine (Nature 2010, 464, 1329). Unlike some other catalysts, which can require organic acids or solvents to function, the molybdenum oxo complex can produce H2 from neutral water solutions and even from seawater.
The catalyst’s ligand can also be more readily and extensively modified than those of other catalysts. That feature “will permit the relationships between catalyst geometric and electronic structure and catalyst performance to be assessed effectively,” says Craig L. Hill, a chemistry professor at Emory University. “Such assessment is key to further improvement in the catalytic rates.”
Another group is aiming to avoid metals entirely in H2-generating systems. A team led by Bjorn Winther-Jensen, a research fellow at the Australian Center for Electromaterials Science at Monash University, prepared an electrode from a porous membrane of polytetrafluoroethylene (Gore-Tex) coated with a layer of poly(3,4-ethylenedioxythiophene) (PEDOT) and polyethylene glycol (PEG) (Adv. Mater. 2010, 22, 1727).
The researchers found that the system could generate H2 from an acidic electrolyte solution at rates comparable with those from systems using traditional Pt-based catalysts, but the polymer composites are both less expensive and more resistant to poisoning than Pt catalysts.
Mechanistically, Winther-Jensen and colleagues believe, the PEG coordinates H3O+ and traps it near the PEDOT, where the reduction to H2 occurs. The porous membrane serves to provide a large surface area for the various components to interact. The system can run for more than 80 days without deteriorating.
The PEDOT-PEG system is “the most active and, undoubtedly, the cheapest catalyst reported so far for H2 evolution from water,” says G. Charles Dismukes, a chemistry professor at Rutgers University. “This opens a route to totally eliminate any metal as catalyst for producing H2 from water.”



Join the conversation
Contact the reporter
Submit a Letter to the Editor for publication
Engage with us on Twitter