Advertisement
Grab your lab coat. Let's get started
Welcome!
Welcome!
Create an account below to get 6 C&EN articles per month, receive newsletters and more - all free.
It seems this is your first time logging in online. Please enter the following information to continue.
As an ACS member you automatically get access to this site. All we need is few more details to create your reading experience.
Not you? Sign in with a different account.
Not you? Sign in with a different account.
ERROR 1
ERROR 1
ERROR 2
ERROR 2
ERROR 2
ERROR 2
ERROR 2
Password and Confirm password must match.
If you have an ACS member number, please enter it here so we can link this account to your membership. (optional)
ERROR 2
ACS values your privacy. By submitting your information, you are gaining access to C&EN and subscribing to our weekly newsletter. We use the information you provide to make your reading experience better, and we will never sell your data to third party members.
Biological Chemistry
Mimic Aids Study Of Protein Acetylation
Derivatizing cysteines to create a mimic of acetyl-lysine groups could be useful in biochemistry
by Stuart A. Borman
July 19, 2010
| A version of this story appeared in
Volume 88, Issue 29
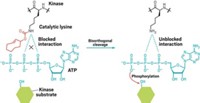
Like protein phosphorylation, acetylation of lysine residues on proteins may have an important regulatory role in cell pathways. Thousands of mammalian proteins are naturally acetylated, but it’s technically difficult to acetylate lysine groups in the lab, complicating the study of the functional consequences of acetylation. Philip A. Cole of Johns Hopkins University School of Medicine and coworkers have now developed an easier method: derivatizing cysteines with methylthiocarbonyl-aziridine (MTCA) to create a thiocarbamate mimic of acetyl-lysine groups (J. Am. Chem. Soc., DOI: 10.1021/ja103954u). This modification is recognized by acetyl-lysine-specific antibodies and by the Brdt bromodomain, a protein receptor that binds acetylated lysines, suggesting that MTCA-modified cysteine is a good stand-in for acetyl-lysine. Unlike acetyl-lysine, the thiocarbamate mimic can’t be deacetylated enzymatically, a property that could make it easier to determine function in deacetylase-rich cell environments. Cole and coworkers demonstrated the utility of the mimic by using it to activate a histone acetyltransferase and a protein kinase, enzymes that are believed to regulate a wide range of cellular processes. The simple method “could be quite useful in biochemistry,” Cole believes.


Join the conversation
Contact the reporter
Submit a Letter to the Editor for publication
Engage with us on Twitter