Advertisement
Grab your lab coat. Let's get started
Welcome!
Welcome!
Create an account below to get 6 C&EN articles per month, receive newsletters and more - all free.
It seems this is your first time logging in online. Please enter the following information to continue.
As an ACS member you automatically get access to this site. All we need is few more details to create your reading experience.
Not you? Sign in with a different account.
Not you? Sign in with a different account.
ERROR 1
ERROR 1
ERROR 2
ERROR 2
ERROR 2
ERROR 2
ERROR 2
Password and Confirm password must match.
If you have an ACS member number, please enter it here so we can link this account to your membership. (optional)
ERROR 2
ACS values your privacy. By submitting your information, you are gaining access to C&EN and subscribing to our weekly newsletter. We use the information you provide to make your reading experience better, and we will never sell your data to third party members.
Physical Chemistry
Covering Up For A Clear View
Surface Science: A blanket of graphene helps reveal the structure of water lying underneath
by Mitch Jacoby
September 6, 2010
| A version of this story appeared in
Volume 88, Issue 36

If a tiny speck of sample contaminant can disturb an ultrasensitive scanning-probe experiment, imagine what burying a sample under a whole sheet of foreign material will do to the measurement. It turns out that when the sample under investigation is water, “burying” it under a sheet of graphene actually helps reveal its atomic-scale structure, according to researchers at California Institute of Technology (Science 2010, 329, 1188).
Much of what’s known about the structure of individual layers of water molecules comes from indirect sample-averaging methods and scanning tunneling microscopy measurements at low temperature, at which water sits still enough for its structure to be probed. Room-temperature measurements have remained challenging because under those conditions water adsorbs weakly to surfaces and its structure is easily altered by probe tips.
The Caltech study describes a novel strategy for studying the nanoscale structure of water in contact with surfaces at room temperature, which is the natural state of affairs for many basic processes in corrosion and in atmospheric and geological chemistry.
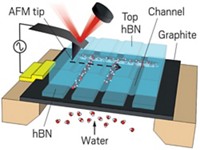
To get a close-up view of adsorbed water layers, Caltech chemists Ke Xu, Peigen Cao, and James R. Heath deposited monolayer graphene sheets onto mica at room temperature under various conditions of relative humidity and analyzed the samples via atomic force microscopy. The group reports that for experiments conducted at 40% relative humidity, they observe numerous islandlike features of various sizes with atomically flat plateaus and uniform heights of roughly 0.37 nm.
By comparing those results with data from various control experiments that account for folded monolayer graphene films, bilayer graphene, and other variables and by repeating the measurements at higher relative humidity, the group came to the conclusion that the structures they observe are islands of water that grow epitaxially on mica and adopt a common ice structure.
The researchers note that the results for the layer of water on the mica surface agree closely with the “puckered bilayer” structure of ice. A second water layer, also adopting an ice structure, forms on top of the layer beneath it when the relative humidity is boosted to near 90%, they say. Subsequent water layers take on a liquidlike structure. The group also reports that the first and second water layers nucleate at nanometer-scale surface defect sites.
In a commentary about the study, Mikhail I. Katsnelson, a condensed-matter theoretician at Radboud University of Nijmegen, in the Netherlands, describes the work as “a breakthrough in studies of the physics and chemistry of wetting.” He notes that covering the water with graphene “protects the water layer and does not disturb its structure.” That conclusion follows from noting that due to graphene’s hydrophobicity, water does not adsorb onto graphene except at cryogenic temperatures. Graphene’s noninterfering role is further substantiated by agreement of the Caltech study’s direct results with results from numerous indirect studies.
The Caltech work is the first application of graphene as a material intended to protect a sample from effects of measuring devices, Katsnelson comments. “The ability to understand wetting at atomic resolution is the first demonstration of the importance of this approach,” he adds. “Others are sure to follow.”



Join the conversation
Contact the reporter
Submit a Letter to the Editor for publication
Engage with us on Twitter