Advertisement
Grab your lab coat. Let's get started
Welcome!
Welcome!
Create an account below to get 6 C&EN articles per month, receive newsletters and more - all free.
It seems this is your first time logging in online. Please enter the following information to continue.
As an ACS member you automatically get access to this site. All we need is few more details to create your reading experience.
Not you? Sign in with a different account.
Not you? Sign in with a different account.
ERROR 1
ERROR 1
ERROR 2
ERROR 2
ERROR 2
ERROR 2
ERROR 2
Password and Confirm password must match.
If you have an ACS member number, please enter it here so we can link this account to your membership. (optional)
ERROR 2
ACS values your privacy. By submitting your information, you are gaining access to C&EN and subscribing to our weekly newsletter. We use the information you provide to make your reading experience better, and we will never sell your data to third party members.
Biological Chemistry
‘Ethical’ Stem Cell Abnormalities Could Hinder Research
New findings reveal that induced pluripotent stem cells can have problematic mutations
by Sarah Everts
March 7, 2011
| A version of this story appeared in
Volume 89, Issue 10
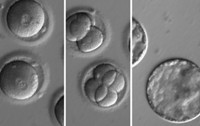
Induced pluripotent stem cells (iPSCs) generated by reprogramming differentiated adult cells are often touted as an “ethical” alternative to embryonic stem cells for applications in regenerative medicine. But research is showing that these cells can have abnormalities that might hinder their eventual clinical development. Two papers in Nature report that techniques for reprogramming fibroblasts into iPSCs cause a 10-fold increase in point mutations and a swell of copy number variations in which sections of DNA are aberrantly repeated (DOI: 10.1038/nature09805 and 10.1038/nature09871). These findings add to three other papers published in recent months reporting potential problems with iPSCs, including chromosomal abnormalities such as trisomies, a common example of which is Down syndrome, as well as incorrect epigenetic marks (Cell Stem Cell, DOI: 10.1016/j.stem.2010.07.017 and 10.1016/j.stem.2010.12.003; Nature, DOI: 10.1038/nature09798). These studies “raise concerns over the implications of such aberrations for future applications of iPSCs,” stem cell researcher Martin F. Pera of the University of Southern California writes in a Nature commentary. Figuring out precisely what in the reprogramming methods causes the genetic and epigenetic abnormalities could permit researchers to find better routes to iPSCs.


Join the conversation
Contact the reporter
Submit a Letter to the Editor for publication
Engage with us on Twitter