Advertisement
Grab your lab coat. Let's get started
Welcome!
Welcome!
Create an account below to get 6 C&EN articles per month, receive newsletters and more - all free.
It seems this is your first time logging in online. Please enter the following information to continue.
As an ACS member you automatically get access to this site. All we need is few more details to create your reading experience.
Not you? Sign in with a different account.
Not you? Sign in with a different account.
ERROR 1
ERROR 1
ERROR 2
ERROR 2
ERROR 2
ERROR 2
ERROR 2
Password and Confirm password must match.
If you have an ACS member number, please enter it here so we can link this account to your membership. (optional)
ERROR 2
ACS values your privacy. By submitting your information, you are gaining access to C&EN and subscribing to our weekly newsletter. We use the information you provide to make your reading experience better, and we will never sell your data to third party members.
Synthesis
Alfred Bader Award In Bioinorganic Or Bioorganic Chemistry
Sponsored by the Alfred Bader Fund
by Stu Borman
January 10, 2011
| A version of this story appeared in
Volume 89, Issue 2
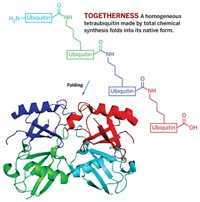
“For developing chemical protein synthesis based on modern ligation methods and for applying total synthesis to the elucidation of the molecular basis of protein function,” chemistry professor Stephen B. H. Kent of the University of Chicago receives this year’s Alfred Bader Award.
In 1992, Kent and a coworker published a paper that pioneered the concept of chemical ligation, the joining of unprotected peptide segments to synthesize protein analogs of a size not previously accessible by chemical synthesis. They demonstrated the technique by using it to synthesize fully active HIV protease from two synthetic peptide building blocks (Science, DOI: 10.1126/science.1566069).
Two years later, Kent and colleagues extended that approach by developing “native chemical ligation,” the joining of unprotected peptides through amide bonds to form totally native proteins of a size difficult or impossible to synthesize in other ways. Native chemical ligation begins with the reaction of an unprotected synthetic peptide thioester with another unprotected peptide containing an amino-terminal cysteine. The resulting thioester-linked intermediate then undergoes a spontaneous intramolecular reaction to form a native peptide bond at the ligation site.
“This second generation ligation chemistry dramatically increases the size of native backbone polypeptides directly accessible by total chemical synthesis,” Kent and coworkers wrote (Science, DOI: 10.1126/science.7973629). “It can be usefully applied to a wide range of synthetic targets, including proteins of moderate size, and it allows direct access to protein functional domains. Native chemical ligation is a foundation stone of a general modular approach to the total chemical synthesis of proteins. ... We believe that general synthetic access of this type will allow almost unlimited variation of the covalent structure of the protein molecule.”
Indeed, since its conception, native chemical ligation has become the most effective chemistry for the covalent condensation of unprotected peptide segments. “The native chemical ligation paper itself has been cited more than 1,000 times, with a citation rate that is still accelerating,” notes Hisashi Yamamoto, Kent’s colleague at the University of Chicago. “Chemical ligation was a breakthrough in protein science and has for the first time led to practical, reproducible, general, and robust synthetic access to the world of proteins. It has been widely applied to enable the application of chemistry to the elucidation of the molecular basis of protein function.”
In the past few years, Kent and coworkers have pioneered the use of mirror-image protein molecules to determine crystal structures of difficult-to-analyze proteins by racemic and quasi-racemate crystallography. And they have developed a route for the first efficient total chemical synthesis of human insulin. Today, Kent’s group continues to refine and optimize chemical techniques for making proteins.
Kent, who is 65, will present the award address before the Division of Biological Chemistry.



Join the conversation
Contact the reporter
Submit a Letter to the Editor for publication
Engage with us on Twitter