Advertisement
Grab your lab coat. Let's get started
Welcome!
Welcome!
Create an account below to get 6 C&EN articles per month, receive newsletters and more - all free.
It seems this is your first time logging in online. Please enter the following information to continue.
As an ACS member you automatically get access to this site. All we need is few more details to create your reading experience.
Not you? Sign in with a different account.
Not you? Sign in with a different account.
ERROR 1
ERROR 1
ERROR 2
ERROR 2
ERROR 2
ERROR 2
ERROR 2
Password and Confirm password must match.
If you have an ACS member number, please enter it here so we can link this account to your membership. (optional)
ERROR 2
ACS values your privacy. By submitting your information, you are gaining access to C&EN and subscribing to our weekly newsletter. We use the information you provide to make your reading experience better, and we will never sell your data to third party members.
Physical Chemistry
Probing The Anomeric Effect
Carbohydrate Chemistry: Peptide sensor provides clues to phenomenon that stabilizes sugars
by Celia Henry Arnaud
January 10, 2011
| A version of this story appeared in
Volume 89, Issue 2

A new sensor probes a well-known, but poorly understood, effect in carbohydrate chemistry that controls the conformations of many biologically relevant sugars. The work provides experimental evidence for the electronic interactions that underlie the so-called anomeric effect.
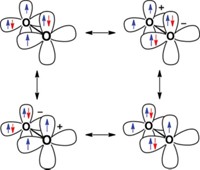
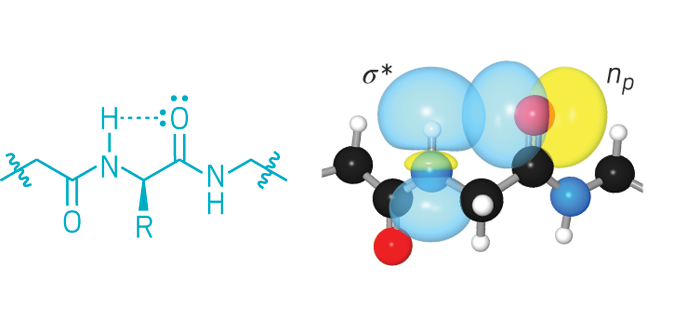
In the anomeric effect, a sugar ring is stabilized by an electronegative substituent at the C1 carbon, also known as the anomeric center. The stabilization is thought to result from interactions between lone electron pairs on oxygen and a C1 antibonding orbital. The effect is called endo-anomeric when the lone pair comes from the oxygen atom in the sugar ring and exo-anomeric when the lone pair comes from oxygen in a substituent on C1.
Benjamin G. Davis, John P. Simons, and coworkers at the University of Oxford use a peptide as a sensor to study the gas-phase anomers of methyl d-galactopyranoside (Nature, DOI: 10.1038/nature09693). The two ends of the peptide form hydrogen bonds with different regions of the sugar. The infrared spectrum reveals information about the shape and bonding of each peptide-anomer complex, Davis says.
In both peptide-anomer complexes, the peptide comes close enough to the sugar’s endocyclic oxygen to sense the electronic density there. Davis’ team concluded that, for both anomers, the exo-anomeric effect is stronger than the endo-anomeric effect.
Their experimental measurements and calculations highlight the importance of the substituent at C2, Davis notes. “The substituent at position 2 can have an enormous influence on the orientation of the bonds at the anomeric center and strongly modulate the exo-anomeric effect,” he says. The C2 position is one that often is modified in naturally occurring sugars, he says.
Vernon G. S. Box, an emeritus chemistry professor at the City College of New York, points out that other sugars such as glycosyl esters are known to have stronger anomeric effects than methyl-glycosides. “It would be nice if their work was extended to the glycosyl esters and glycosyl halides to provide more insight into and evidence for a truly valid rationalization of what is observed experimentally,” he says.


Join the conversation
Contact the reporter
Submit a Letter to the Editor for publication
Engage with us on Twitter