Advertisement
Grab your lab coat. Let's get started
Welcome!
Welcome!
Create an account below to get 6 C&EN articles per month, receive newsletters and more - all free.
It seems this is your first time logging in online. Please enter the following information to continue.
As an ACS member you automatically get access to this site. All we need is few more details to create your reading experience.
Not you? Sign in with a different account.
Not you? Sign in with a different account.
ERROR 1
ERROR 1
ERROR 2
ERROR 2
ERROR 2
ERROR 2
ERROR 2
Password and Confirm password must match.
If you have an ACS member number, please enter it here so we can link this account to your membership. (optional)
ERROR 2
ACS values your privacy. By submitting your information, you are gaining access to C&EN and subscribing to our weekly newsletter. We use the information you provide to make your reading experience better, and we will never sell your data to third party members.
Analytical Chemistry
Extreme Photoswitching
Microscopy: Fluorescent proteins that turn on and off many more times than usual improve superresolution methods
by Celia Henry Arnaud
September 19, 2011
| A version of this story appeared in
Volume 89, Issue 38

Two new photoswitchable fluorescent proteins that can be repeatedly turned on and off extend the capabilities of superresolution fluorescence microscopy, German researchers report in a pair of papers. The proteins may find use in biological imaging and data storage.
Stefan W. Hell, Stefan Jakobs, and coworkers at the Max Planck Institute for Biophysical Chemistry, in Göttingen, mutated existing fluorescent proteins to generate the new proteins. Starting with a yellow fluorescent protein called Citrine, they found a protein, dubbed Dreiklang, which can be switched between fluorescent and nonfluorescent states as many as 200 times (Nat. Biotechnol., DOI: 10.1038/nbt.1952). Most other fluorescent proteins can generally be switched on and off only about 20 times.
Dreiklang’s other distinguishing feature is the fact that different wavelengths are used to excite fluorescence and to switch the protein to its dark state. This, together with the long lifetime of the on and off states, allows scientists to use lower intensity light for biological imaging and thereby do less damage to tissue.
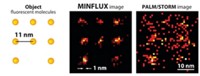
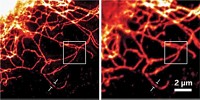
Using a similar strategy, the researchers developed a second protein, reversibly switchable enhanced green fluorescent protein (rsEGFP). And they show that rsEGFP can be used to write and read data using the superresolution microscopy method known as RESOLFT (Nature, DOI: 10.1038/nature10497). In RESOLFT, patterned light turns off emission everywhere except in a precisely defined area. By moving the pattern slightly and repeating the process, the researchers can make closely spaced features. They are able to do this only because rsEGFP can be switched more than 1,000 times.
Writing requires that the data spots stay off. In addition to its reversible off state, rsEGFP also has a permanent off state. The protein switches on with 405-nm light, reversibly turns off with 491-nm light, and permanently turns off with 532-nm light. The features made this way are so small and so close together that they can be read only with superresolution microscopy.
Both of the proteins can be used as fluorescent probes for superresolution imaging in live cells by expressing them as fusions with other proteins.
“These novel photoswitchable fluorescent probes are remarkable in that they can switch between fluorescent and dark states for hundreds to thousands of cycles,” says Xiaowei Zhuang, a chemist at Harvard University who also develops superresolution imaging techniques. “This incredible level of fatigue resistance has enabled live-cell imaging with low light intensity and minimal phototoxicity. These advances represent a major breakthrough in superresolution fluorescence microscopy and substantially enhance our ability to resolve subcellular structures and dynamics.”


Join the conversation
Contact the reporter
Submit a Letter to the Editor for publication
Engage with us on Twitter