Advertisement
Grab your lab coat. Let's get started
Welcome!
Welcome!
Create an account below to get 6 C&EN articles per month, receive newsletters and more - all free.
It seems this is your first time logging in online. Please enter the following information to continue.
As an ACS member you automatically get access to this site. All we need is few more details to create your reading experience.
Not you? Sign in with a different account.
Not you? Sign in with a different account.
ERROR 1
ERROR 1
ERROR 2
ERROR 2
ERROR 2
ERROR 2
ERROR 2
Password and Confirm password must match.
If you have an ACS member number, please enter it here so we can link this account to your membership. (optional)
ERROR 2
ACS values your privacy. By submitting your information, you are gaining access to C&EN and subscribing to our weekly newsletter. We use the information you provide to make your reading experience better, and we will never sell your data to third party members.
People
Herbert C. Brown Award For Creative Research In Synthetic Methods
Sponsored by the Purdue Borane Research Fund and the Herbert C. Brown Award Endowment
by Cheryl Hogue
February 21, 2011
| A version of this story appeared in
Volume 89, Issue 8
Akira Suzuki published a key paper in 1979, the same year his postdoctoral mentor, Herbert C. Brown, won the Nobel Prize in Chemistry for work with organoboranes. In that paper, Suzuki and his colleague Norio Miyaura reported a palladium-catalyzed coupling of organoboron compounds with organohalides (Tetrahedron Lett., DOI: 10.1016/s0040-4039(01)95429-2).
That seminal work, which has led to numerous innovations in organic chemical synthesis, earned Suzuki his own Nobel Prize in 2010. Suzuki shared the award with two others chemists for their work on palladium-catalyzed cross-couplings in organic synthesis (C&EN, Oct. 11, 2010, page 7).
Suzuki, 80, is an emeritus professor of chemistry at Japan’s Hokkaido University. As a young man, Suzuki was attracted to mathematics and considered majoring in that subject. But his interests shifted when he learned organic chemistry using Louis F. and Mary Fieser’s textbook “Organic Chemistry.”
“I decided to specialize in organic synthesis,” Suzuki tells C&EN. He earned undergraduate and doctoral degrees from Hokkaido, where he became an associate professor in 1961.
From 1963 to 1965, Suzuki worked at Purdue University with Brown, one of the 20th century’s most prominent chemists. Suzuki says he was greatly influenced by systematic study of Brown’s thinking.
In addition, Brown encouraged Suzuki to focus on research that would have major implications for the field of chemistry. “He told me often, ‘You have to have work which is quoted in textbooks,’ ” Suzuki says. “This is one of my mottoes.”
After working in Brown’s laboratory, Suzuki returned to Hokkaido and studied new methods of synthesis using organic boron compounds. He followed Brown’s advice. The coupling described in the 1979 paper—known as the Suzuki reaction—is widely quoted in chemistry textbooks.
The reaction led to major advances in organic chemistry. The method allows researchers to precisely place aromatic rings in novel compounds, explains John A. Soderquist, professor of chemistry at the University of Puerto Rico, Río Piedras.
“It changed the direction of pharmaceutical chemistry dramatically,” Soderquist tells C&EN. For example, the Suzuki reaction has led to an increased number of aromatic-to-aromatic bonds in compounds that are candidates for new drugs, he says.
Materials science has also applied the Suzuki coupling. Researchers have used it to attach light-sensing molecular components to nanoparticles, Soderquist says.
“It has clearly revolutionized the way we look at the construction of carbon-carbon bonds,” Soderquist continues. Noteworthy features of this process include forming carbon-carbon bonds with a variety of organoboranes and carbon electrophiles as combining partners. “Significantly, organoboranes can be prepared by a variety of methods including organometallic methods, hydroboration, and haloboration,” Soderquist says.
Haloboration was an obscure reaction that Suzuki developed into an important new path to functionalized vinylboranes, Soderquist says. “The intermediate organoboranes have low toxicity and are stable, often isolable reagents which permit many functional groups to be tolerated in the coupling process.”
Soderquist points out that Suzuki didn’t patent the coupling. “He’s never made a dime from this reaction,” Soderquist says, commenting on Suzuki’s “refreshing lack of egotism.”
Suzuki will present the award address before the Division of Organic Chemistry.



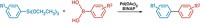


Join the conversation
Contact the reporter
Submit a Letter to the Editor for publication
Engage with us on Twitter