Advertisement
Grab your lab coat. Let's get started
Welcome!
Welcome!
Create an account below to get 6 C&EN articles per month, receive newsletters and more - all free.
It seems this is your first time logging in online. Please enter the following information to continue.
As an ACS member you automatically get access to this site. All we need is few more details to create your reading experience.
Not you? Sign in with a different account.
Not you? Sign in with a different account.
ERROR 1
ERROR 1
ERROR 2
ERROR 2
ERROR 2
ERROR 2
ERROR 2
Password and Confirm password must match.
If you have an ACS member number, please enter it here so we can link this account to your membership. (optional)
ERROR 2
ACS values your privacy. By submitting your information, you are gaining access to C&EN and subscribing to our weekly newsletter. We use the information you provide to make your reading experience better, and we will never sell your data to third party members.
Nanomaterials
New Method Reveals Nanoparticle Solubility
Nanomaterials: Researchers examine nanoparticle degradation in salt solutions
by Lauren K. Wolf
December 21, 2011

Scientists studying how nanomaterials behave in the environment have developed a new technique for assessing how quickly the tiny particles dissolve in aqueous solutions (Environ. Sci. Technol., DOI: 10.1021/es203475a). The method’s developers hope it will help scientists predict the effects of nanoparticles on the environment and design safer materials.
Manufacturers incorporate silver nanomaterials into consumer products such as paints and clothing because of the tiny particles’ ability to kill bacteria. One explanation for the nanoparticles’ antimicrobial effectiveness is that they shed toxic silver ions when they dissolve. Environmental scientists worry that when these materials reach the environment and start to dissolve, the release of silver could harm aquatic ecosystems.
Previous studies on how nanoparticles dissolve have not separated the behavior of individual particles from that of aggregated clumps. Just as snow melts more slowly when packed into a mass, nanoparticles dissolve more slowly when clustered. Environmental engineers Peter J. Vikesland and Ronald D. Kent of Virginia Polytechnic Institute & State University wanted to watch how the particles dissolved when isolated from each other to establish an upper limit on how quickly they could shed ions.
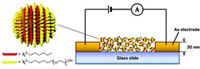
To prevent the materials from aggregating, the researchers used lithography to affix silver nanoparticles at intervals on a glass surface. They then exposed the evenly spaced nanoparticles to salt solutions of varying concentration for two weeks and used atomic force microscopy to probe changes to their size and shape.
At the highest concentration of sodium chloride Vikesland and Kent explored—a level similar to that of seawater—they observed an almost 50% reduction in both the height and width of the tetrahedral-shaped silver particles. At the lowest salt concentration—one resembling the chloride levels inside cells—the pair saw less than a 5% change in the particles’ size. The results suggest, the researchers say, that chloride accelerates the shedding of silver ions from the particles.
Vikesland admits that he’s not yet sure how the chloride catalyzes the conversion of silver atoms to silver ions. But data that he and Kent collected by X-ray photoelectron spectroscopy show that silver chloride does not form.
Bernd Nowack, an environmental chemist at the Swiss Federal Laboratories for Materials Science & Technology says the method’s biggest advantage is that it allows researchers to study silver nanoparticles at very low concentrations, similar to those present in the environment. “In fact,” he says, “I can envisage it eventually being used directly in the environment, in rivers or wastewater, for instance.”
Aside from looking at the effects on nanoparticle stability of more-complex, realistic water samples, Vikesland says his group will next investigate the influence of particle coatings. They want to test the idea that these protective layers of organic macromolecules can hinder particle dissolution.


Join the conversation
Contact the reporter
Submit a Letter to the Editor for publication
Engage with us on Twitter