Advertisement
Grab your lab coat. Let's get started
Welcome!
Welcome!
Create an account below to get 6 C&EN articles per month, receive newsletters and more - all free.
It seems this is your first time logging in online. Please enter the following information to continue.
As an ACS member you automatically get access to this site. All we need is few more details to create your reading experience.
Not you? Sign in with a different account.
Not you? Sign in with a different account.
ERROR 1
ERROR 1
ERROR 2
ERROR 2
ERROR 2
ERROR 2
ERROR 2
Password and Confirm password must match.
If you have an ACS member number, please enter it here so we can link this account to your membership. (optional)
ERROR 2
ACS values your privacy. By submitting your information, you are gaining access to C&EN and subscribing to our weekly newsletter. We use the information you provide to make your reading experience better, and we will never sell your data to third party members.
Synthesis
Easier Path To Leading Antimalarial Drug
Continuous-flow strategy for scaling up natural product synthesis could bring down the cost of the drug artemisinin
by Bethany Halford
December 24, 2012
| A version of this story appeared in
Volume 90, Issue 52


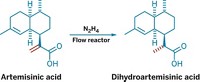
Gaining access to the antimalarial compound artemisinin became potentially easier and less expensive this year when researchers in Germany announced that they developed a continuous-flow semisynthetic procedure for making the drug. Their strategy could be applied to the complex syntheses of other natural products (C&EN, Jan. 23, page 4; Angew. Chem. Int. Ed., DOI: 10.1002/anie.201107446). Artemisinin is currently the most effective treatment in the fight against multi-drug-resistant forms of malaria. Although artemisinin can be extracted from Artemisia annua, the herbaceous plant is seasonal, making the supply unreliable and the cost volatile. By next year, drugmaker Sanofi plans to produce 40 tons of artemisinin via a semisynthetic route based on the intermediate artemisinic acid—a compound that can be produced by a specially engineered yeast. But converting artemisinic acid to artemisinin is no simple task. It involves installing an endoperoxide group and generating three of artemisinin’s rings. Doing this complex chemistry on a scale sufficient to meet the demand of 225 million malaria patients in developing countries presents a formidable challenge. That’s where the work of Peter H. Seeberger and François Lévesque comes in. The researchers, from the Max Planck Institute for Colloids & Interfaces, figured out how to combine three key reactions into a single continuous-flow process that converts dihydroartemisinic acid—made by reducing artemisinic acid—to artemisinin. Since its report in January, Seeberger’s group has optimized the process, upping the yield from 39% to 65%, which allows them to purify the product via crystallization rather than chromatography. The ultimate impact of the continuous-flow process remains to be seen. Sanofi has no plan to adopt it for artemisinin production, but Seeberger has set up a company, ArtemiFlow, in Potsdam, Germany, aimed at making the drug. Seeberger and coworkers are in the process of finishing a reactor that will produce more than 1 metric ton of artemisinin per year, he tells C&EN. The company is also developing a 10-ton reactor.


Join the conversation
Contact the reporter
Submit a Letter to the Editor for publication
Engage with us on Twitter