Advertisement
Grab your lab coat. Let's get started
Welcome!
Welcome!
Create an account below to get 6 C&EN articles per month, receive newsletters and more - all free.
It seems this is your first time logging in online. Please enter the following information to continue.
As an ACS member you automatically get access to this site. All we need is few more details to create your reading experience.
Not you? Sign in with a different account.
Not you? Sign in with a different account.
ERROR 1
ERROR 1
ERROR 2
ERROR 2
ERROR 2
ERROR 2
ERROR 2
Password and Confirm password must match.
If you have an ACS member number, please enter it here so we can link this account to your membership. (optional)
ERROR 2
ACS values your privacy. By submitting your information, you are gaining access to C&EN and subscribing to our weekly newsletter. We use the information you provide to make your reading experience better, and we will never sell your data to third party members.
Biological Chemistry
On The Origin Of Sugars
Prebiotic Chemistry: Experiments reveal a possible path to forming the sugar in RNA
by Melissae Fellet
January 27, 2012

New experiments strengthen a theory about how sugars could have formed on early Earth (J. Am. Chem. Soc., DOI: 10.1021/ja211383c). This set of reactions could help uncover the nucleic acid predecessor to RNA, scientists say.
The sugar ribose forms the backbone of RNA, the coworker--and nucleic acid ancestor--of DNA. Scientists trying to imagine how life arose from chemicals on Earth have looked to the formose reaction to build the sugar portion of RNA from formaldehyde. This reaction creates a mixture of sugars with varying sizes and shapes, of which ribose makes up less than 1%. So chemists have experimented with different likely prebiotic conditions to control the reaction and build more ribose.
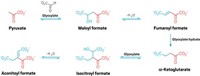
In 2007, Albert Eschenmoser, now retired from Scripps Research Institute in California, proposed a different set of reactions to build sugars in a prebiotic world, starting with the two-carbon molecule glyoxylate and its dimer dihydroxyfumarate (DHF) (Tetrahedron, DOI: 10.1016/j.tet.2007.10.012). Intermediates could also lead to other biological building blocks like α-amino acids and pyrimidines.
Now Ram Krishnamurthy and his colleagues at Scripps Research Institute have tested whether some of these reactions produce the predicted sugars. The researchers reacted the dilithium salt of DHF with simple aldehydes like glyoxylate, glycolaldehyde, and glyceraldehyde. The reaction with glyceraldehyde produced a mixture of the five-carbon sugars xylulose and ribulose with 60% yield. The researchers also detected intermediates by 13C NMR that could lead to α-amino acids in other reactions.
“We were surprised at how clean and efficient the reactions were,” Krishnamurthy says. The DHF reactions gave few side products, he explains, and left little starting material behind. The researchers are now testing the reactions under plausible prebiotic conditions, such as using different DHF salts at low temperatures and concentrations. But for Eschenmoser’s “glyoxylate scenario” to be a possible route to the first sugars, a large challenge remains: Scientists have never built the starting material DHF under conditions that mimic those on early Earth.
Nonetheless, building ribulose so cleanly is significant, says George Cody, an organic geochemist at the Carnegie Institution for Science. Prebiotic conditions may have existed that transformed ribulose to ribose without the enzymes that do that job in our bodies today, he notes, although the reactions are still unknown.
This “really nice chemistry” provides motivation to look for ways to make DHF in model prebiotic reactions, says Nicholas Hud, a chemist at Georgia Institute of Technology.
If researchers find a plausible way to make DHF, Eschenmoser’s scenario could provide clues to which nucleic acid preceded RNA, he says. Because these reactions form ribulose so efficiently, that sugar–and not ribose–may have been one of the earliest sugars, Hud says. “When I look at ribose, it seems like a product of evolution,” he adds. “It’s hard to make under prebiotic conditions, and it’s perfect for what it does today.”


Join the conversation
Contact the reporter
Submit a Letter to the Editor for publication
Engage with us on Twitter