Advertisement
Grab your lab coat. Let's get started
Welcome!
Welcome!
Create an account below to get 6 C&EN articles per month, receive newsletters and more - all free.
It seems this is your first time logging in online. Please enter the following information to continue.
As an ACS member you automatically get access to this site. All we need is few more details to create your reading experience.
Not you? Sign in with a different account.
Not you? Sign in with a different account.
ERROR 1
ERROR 1
ERROR 2
ERROR 2
ERROR 2
ERROR 2
ERROR 2
Password and Confirm password must match.
If you have an ACS member number, please enter it here so we can link this account to your membership. (optional)
ERROR 2
ACS values your privacy. By submitting your information, you are gaining access to C&EN and subscribing to our weekly newsletter. We use the information you provide to make your reading experience better, and we will never sell your data to third party members.
Biological Chemistry
Single Gene Allows Scientists To Track Cells In Multiple Ways
Imaging: Cells spotted on MRI, fluorescence imaging, and nuclear-medicine scans
by Katherine Bourzac
March 15, 2012

Tracking cells in the body is a key, but challenging, step in developing gene and cell therapies. Now researchers have developed a way to make cells visible to many different types of imaging methods, including magnetic resonance imaging (MRI), fluorescence imaging, and single-photon emission computed tomography (J. Am. Chem. Soc., DOI: 10.1021/ja209868g). They hope the method will help monitor therapies in the clinic and enable in-depth studies of diseases including cancer.
A handful of early-stage trials of gene therapy are under way in the U.S. For example, in some trials, researchers take cancer patients’ T cells and use viruses to insert genes that they hope will enable the patients’ immune systems to recognize and attack the cancer. But when such an immunotherapy fails in the clinic, it can be difficult to uncover why. “There’s no way of finding how many [treated] cells live, are they going to the tumor site, are they multiplying,” says Bakhos Tannous, a neuroscientist at Harvard Medical School.
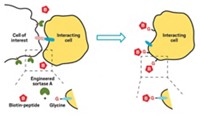
To track cells inside the body, researchers can give the cells a “reporter” gene. For example, in mice researchers can track cells using reporter genes that encode fluorescent proteins and bioluminescent proteins. But these reporters don’t work in the clinic: Light from the proteins scatters before it can travel out of the body from tissues below the surface. Another approach to making reporters is to create a target that alone may not be visible on imaging scans such as MRI, but that will bind a variety of contrast agents: molecules that light up in those scanning methods. The ideal reporter would be universal, making a cell a target that can be bound by contrast agents for any imaging technique.
Tannous and his colleagues have taken a step in that direction, demonstrating a reporter that can serve as a target for several imaging labels. They designed the reporter gene so that the resulting protein sat on the cell membrane and contained a peptide that an enzyme in cells recognizes and attaches to the vitamin biotin. Cells that make the reporter protein end up decorated with biotin, which makes them a target for any molecule, such as an imaging label, attached to a protein called streptavidin. Biotin and streptavidin have one of the strongest known noncovalent interactions in nature.
To prove that the reporter works for spotting specific cells, the researchers used it with three imaging techniques to monitor the formation and growth of brain tumors in mice. First, they used a virus to add the reporter gene to brain-cancer cells, which they then injected into mice’s brains. Two weeks later they injected the mice with imaging labels bound to streptavidin: For MRI studies, for example, they injected the mice with magnetic nanoparticles bound to streptavidin. With all three imaging methods, the researchers detected signals from the tumor after they injected the labels. In control mice that received brain-cancer cells expressing a reporter gene for a fluorescent protein, none of the labels led to signals from the tumors.
Renier Brentjens, an oncologist at the Memorial Sloan-Kettering Cancer Center, says the reporter shows promise for tracking cells in the clinic. However, he notes that the transmembrane domain of the reporter came from a crustacean, and he says that researchers will need to show that it doesn’t cause an immune reaction in people.


Join the conversation
Contact the reporter
Submit a Letter to the Editor for publication
Engage with us on Twitter