Advertisement
Grab your lab coat. Let's get started
Welcome!
Welcome!
Create an account below to get 6 C&EN articles per month, receive newsletters and more - all free.
It seems this is your first time logging in online. Please enter the following information to continue.
As an ACS member you automatically get access to this site. All we need is few more details to create your reading experience.
Not you? Sign in with a different account.
Not you? Sign in with a different account.
ERROR 1
ERROR 1
ERROR 2
ERROR 2
ERROR 2
ERROR 2
ERROR 2
Password and Confirm password must match.
If you have an ACS member number, please enter it here so we can link this account to your membership. (optional)
ERROR 2
ACS values your privacy. By submitting your information, you are gaining access to C&EN and subscribing to our weekly newsletter. We use the information you provide to make your reading experience better, and we will never sell your data to third party members.
Biological Chemistry
Spring-Loaded Nanoparticles Sense Proteases
Biosensors: A nanoparticle squeezed by peptides could help detect active enzymes associated with disease
by Erika Gebel
April 18, 2012

To spot early signs of cancer, some doctors want to monitor protease activity in the body. Previous research has shown that certain proteases turn on at the early stages of disease. Now researchers have developed a nanoparticle to help: It lights up only around active proteases (J. Am. Chem. Soc., DOI: 10.1021/ja301259v).
As cancer starts developing, few cancer cells exist in the body, so the proteases that accompany them are relatively rare. As a result, encounters are infrequent between the proteases and any chemical sensor designed to detect them. Timothy M. Swager of Massachusetts Institute of Technology wanted to devise a sensor that produced an intense signal so researchers could spot these rare events.
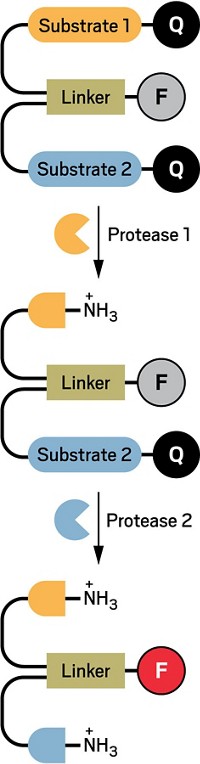
He and his colleagues designed polymer nanoparticles that would act like protease mouse traps. Small peptides on the particle’s surface put strain on the particle’s polymers, keeping them in an “off” state. When a protease snips a few of the peptides, the polymers’ strain releases, producing an “on” state. The researchers wanted the on state to glow brightly so that they could detect signal from as few nanoparticles as possible.
Fortunately, Swager and his team knew of a polymer that could produce such a glow and turn off with strain. The material, which Swager calls “an old friend,” is a polymer based on pentiptycene that emits red light when ultraviolet light excites it. Swager predicted that in a tightly-packed nanoparticle, such as the strained ones he wanted to make, these polymer “chains would mingle together,” which would prevent them from emitting light. If given a bit more room, he says, they should start emitting again. Also, because a nanoparticle would contain multiple strands of this polymer, when a protease activated the sensor, it would trigger many copies of the dye to glow, instead of just one, as in most previous dye-based sensors.
To complete the sensor design, the chemists then added two other polymer blocks to each end of the pentiptycene polymer. One block contained chemical groups they could attach to one end of a peptide. The final block was a hydrophilic one, to prevent the nanoparticles from aggregating in water. The resulting five-block polymer wasn’t “trivial to make,” says Swager. When added to water and sonicated, the polymers formed nanoparticles of about 80 nm in diameter.
To test the particles, the researchers cross-linked the nanoparticles’ polymers with a peptide that had an amino acid sequence that the protease trypsin would cleave. The resulting particles barely glowed. When they mixed the dark nanoparticles with trypsin, the particles began to glow 15 times as brightly as they did without the protease.
Charles S. Craik of the University of California, San Francisco, says the nanoparticle design is a clever way to look for specific proteases. All researchers would have to do to spot other enzymes is change the crosslinking peptide’s sequence.
But he suggests that the researchers improve the particles’ brightness and determine whether enzymes other than proteases can turn them on. Swager agrees that the signal isn’t yet strong enough for a commercial cancer detection system, but he hopes to get there soon.


Join the conversation
Contact the reporter
Submit a Letter to the Editor for publication
Engage with us on Twitter