Advertisement
Grab your lab coat. Let's get started
Welcome!
Welcome!
Create an account below to get 6 C&EN articles per month, receive newsletters and more - all free.
It seems this is your first time logging in online. Please enter the following information to continue.
As an ACS member you automatically get access to this site. All we need is few more details to create your reading experience.
Not you? Sign in with a different account.
Not you? Sign in with a different account.
ERROR 1
ERROR 1
ERROR 2
ERROR 2
ERROR 2
ERROR 2
ERROR 2
Password and Confirm password must match.
If you have an ACS member number, please enter it here so we can link this account to your membership. (optional)
ERROR 2
ACS values your privacy. By submitting your information, you are gaining access to C&EN and subscribing to our weekly newsletter. We use the information you provide to make your reading experience better, and we will never sell your data to third party members.
Energy
A Solid Solar-Cell Solution
Alternative Energy: New solid electrolyte bypasses problems of traditional dye-sensitized solar cells
by Mitch Jacoby
May 31, 2012

By replacing the liquid component of a class of thin-film solar cells with a solid, researchers at Northwestern University have come up with a design that avoids the corrosion and durability problems associated with the liquid variety. The study, reported in a paper in Nature (DOI: 10.1038/nature11067), may lead to new types of inexpensive, long-lasting, and high-performing solar cells composed entirely of solid-state components.

Photovoltaic, or solar-cell, technology holds enormous promise for tapping usable, carbon-free energy from the sun. Some three-quarters of electricity-producing rooftop photovoltaic panels and other types of solar arrays are based on crystalline silicon cells, a well-established but expensive technology.
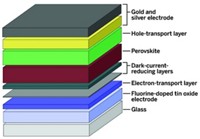
Dye-sensitized solar cells (DSSCs) are one low-cost alternative to crystalline semiconductor photovoltaics. The heart of DSSCs consists of a film of inexpensive porous titanium dioxide nanoparticles coated with a sunlight-absorbing dye, often a ruthenium compound, in contact with a liquid electrolyte.
The most common electrolyte is an organic solution of the iodide/triodide (I–/I3–) redox couple. This material conducts positive charge (or “holes”) and plays a central role in shuttling charge through the cell.
DSSCs are made from inexpensive components and can be fabricated via low-cost manufacturing processes. But their long-term stability and durability is limited by several factors: The electrolyte solution is corrosive, volatile, and prone to leaking, and it can react with the dye and other cell components.
Researchers have shown previously that the liquid electrolyte can be replaced with a noncorrosive solid-state counterpart. Until now, however, the efficiency of solid-state cells in converting sunlight to electricity has been low.
Northwestern chemistry professor Mercouri Kanatzidis and postdoc In Chung have come up with a replacement material for DSSCs that sidesteps the cells’ problems. Specifically, the group, which includes Robert P. H. Chang, Byunghong Lee, and Jiaqing He, has shown that substituting the liquid electrolyte with a new semiconducting inorganic solid—fluorine-doped cesium tin iodide (CsSnI2.95F0.05)—avoids the corrosion and reactivity problems and leads to cells with conversion efficiencies of around 10%. That value is comparable to the efficiencies obtained with newly fabricated DSSCs. They also found that the new cells stand up well to high temperatures, high levels of humidity, and accelerated ageing tests.
Kanatzidis points out that unlike conventional solar-cell electrolytes, CsSnI3 absorbs light strongly and causes the new cells to absorb more of the red portion of the visible spectrum than that captured by traditional DSSCs. That property, which may play an important role in the new cell’s high performance, is not yet understood and is the focus of follow-up studies, he says.
The performance of the new cells is encouraging, says chemistry professor Thomas E. Mallouk of Pennsylvania State University. Mallouk, who wrote a commentary for the same issue of Nature, adds that not only does the cell have low internal electrical resistance, which is to be expected for cells that have good hole conductors, but it also achieves high current and voltage.
Given that a range of solid solutions can be made from compounds in this family, “elaborating the chemistry of this class of hole conductors may be a fruitful avenue for dye-cell research,” he adds.




Join the conversation
Contact the reporter
Submit a Letter to the Editor for publication
Engage with us on Twitter