Advertisement
Grab your lab coat. Let's get started
Welcome!
Welcome!
Create an account below to get 6 C&EN articles per month, receive newsletters and more - all free.
It seems this is your first time logging in online. Please enter the following information to continue.
As an ACS member you automatically get access to this site. All we need is few more details to create your reading experience.
Not you? Sign in with a different account.
Not you? Sign in with a different account.
ERROR 1
ERROR 1
ERROR 2
ERROR 2
ERROR 2
ERROR 2
ERROR 2
Password and Confirm password must match.
If you have an ACS member number, please enter it here so we can link this account to your membership. (optional)
ERROR 2
ACS values your privacy. By submitting your information, you are gaining access to C&EN and subscribing to our weekly newsletter. We use the information you provide to make your reading experience better, and we will never sell your data to third party members.
Pharmaceuticals
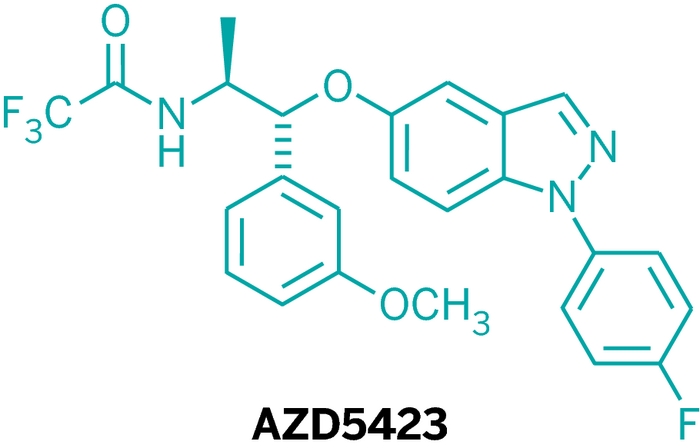
AZD5423, For Chronic Obstructive Pulmonary Disease
by Carmen Drahl
April 22, 2013
| A version of this story appeared in
Volume 91, Issue 16

Collaborations between large pharmaceutical companies usually don’t happen until relatively late in drug development. But the challenge of developing a new drug for respiratory diseases prompted AstraZeneca to collaborate from the get-go. In New Orleans, Thomas Hansson, a senior principal investigator at the company, presented the fruit of international teamwork with Bayer Schering Pharma—AZD5423, a potential medication for chronic obstructive pulmonary disease (COPD).
COVER STORY
AZD5423, For Chronic Obstructive Pulmonary Disease
Inflammation is a major cause of the airway blockages people experience in asthma or COPD. Inhalable glucocorticoids, a class of steroid hormones, can decrease that inflammation and swelling, either alone or in combination with agents that dilate the respiratory tract. The most common cause of COPD is smoking, so quitting can bring relief in some cases.
AZD5423
\
Company: AstraZeneca
Target: glucocorticoid receptor
Disease: chronic obstructive pulmonary disease
Many people with COPD don’t respond to steroids. And long-term steroid use can lead to side effects including increased risk of infections. So Hansson and his colleagues aimed to develop a safer, more efficacious, nonsteroidal COPD treatment.
They targeted the receptor to which glucocorticoids bind. When activated, this receptor helps to regulate a variety of genes, including some linked to inflammation.
Work from many teams had already turned up nonsteroidal molecules that acted on the glucocorticoid receptor. Hansson’s team at AstraZeneca entered into a four-year collaboration with researchers at Bayer Schering Pharma, in Germany, to identify leads of their own with high-throughput screens. It’s difficult to obtain nonsteroidal compounds potent enough to be used in a dry-powder inhaler, so that was one of the team’s biggest challenges, Hansson says.
The team transformed a sulfonamide hit from a screen into molecules that acted on the glucocorticoid receptor efficiently. The molecules set off red flags in a biological test for potential carcinogens, however, because they were forming aromatic amine metabolites that can be toxic to genes. Hansson’s team was able to remove those motifs, “but then the compounds had no anti-inflammatory activity anymore,” he says.
The collaboration with Bayer’s team made it easier to solve that conundrum. The two firms explored multiple chemical avenues in parallel. That effort, coupled with structural and computational chemistry work at AstraZeneca, led to a solution. The researchers decided to work with a completely different class of molecules—indazole ethers—because this resolved the carcinogen issue while still controlling glucocorticoid receptor signaling. After multiple rounds of adjustments, the team tested their most potent compounds in lab animals designed to help approximate human lung disease. AZD5423 performed best in those tests, and was selected to enter human clinical trials.
The compound is now in a Phase II study in patients with COPD, where its efficacy and safety are being measured against that of a typical steroid or a placebo. “It was a lot of fun to work on this project,” Hansson says. “AstraZeneca and Bayer each brought pieces to the puzzle.”




Join the conversation
Contact the reporter
Submit a Letter to the Editor for publication
Engage with us on Twitter