Advertisement
Grab your lab coat. Let's get started
Welcome!
Welcome!
Create an account below to get 6 C&EN articles per month, receive newsletters and more - all free.
It seems this is your first time logging in online. Please enter the following information to continue.
As an ACS member you automatically get access to this site. All we need is few more details to create your reading experience.
Not you? Sign in with a different account.
Not you? Sign in with a different account.
ERROR 1
ERROR 1
ERROR 2
ERROR 2
ERROR 2
ERROR 2
ERROR 2
Password and Confirm password must match.
If you have an ACS member number, please enter it here so we can link this account to your membership. (optional)
ERROR 2
ACS values your privacy. By submitting your information, you are gaining access to C&EN and subscribing to our weekly newsletter. We use the information you provide to make your reading experience better, and we will never sell your data to third party members.
Drug Development
For Five New Drug Candidates, Structures Are Now Public
Potential treatments for cancer, respiratory disease, heart trouble unveiled at ACS meeting
by Carmen Drahl
April 22, 2013
| A version of this story appeared in
Volume 91, Issue 16
Take a conference center ballroom, a crowd of chemists, and drug structures emerging from the shroud of secrecy, and you have the recipe for one of the most consistently popular sessions at the ACS national meeting.
At the spring meeting in New Orleans, the Division of Medicinal Chemistry’s traditional “First-Time Disclosures” session revealed five drug candidate structures, each from a different company. The molecules face a tough road ahead in clinical trials, but they have at least a chance of becoming medications in the future.
Researchers filled the ballroom, furiously scribbling structures on notepads. The session was organized by Albert J. Robichaud, chief scientific officer of Cambridge, Mass.-based Sage Therapeutics.
The talks have always featured excellent work from companies both large and small, Robichaud told C&EN. But the changes to pharma’s landscape over the past five years have translated into more small firms taking the podium. “I think this is a great example of scientists finding opportunities in small organizations after the significant downsizing of large pharma research groups,” he said. Moreover, he added, the shift adds to the ranks of firms doing high-quality science for the benefit of patients.
Where are they now?
An Inflammation Drug Flames Out
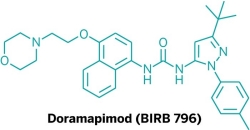
Molecules revealed at the ACS meeting often don’t pan out, despite high hopes. For instance, the road has been rocky for blockers of a kinase enzyme called p38, experimental drugs designed to treat rheumatoid arthritis or Crohn’s disease. At the spring 2002 ACS national meeting, Boehringer Ingelheim debuted the structure of its p38 blocker BIRB 796. But the firm has since pulled the plug on this compound.
Both rheumatoid arthritis and Crohn’s involve chronic inflammation. BIRB 796 was designed to mitigate those symptoms, because the kinase p38 had been implicated in triggering inflammation. After the 2002 symposium, BIRB 796 got a new name—doramapimod. The molecule underwent testing in people with Crohn’s disease, rheumatoid arthritis, and psoriasis.
In Crohn’s, the trial uncovered “no evidence for clinical efficacy” (Clin. Gastroenterol. Hepatol. 2006, 4, 325). Results in psoriasis and rheumatoid arthritis were similarly disappointing, according to Stanford University Medical Center’s Mark C. Genovese, who penned an editorial about the agent in 2009 (Arthritis Rheum., DOI: 10.1002/art.24264). Boehringer Ingelheim ultimately dropped doramapimod because patients on it were developing liver function abnormalities. A 2011 study from a Japanese firm concluded that doramapimod became a reactive epoxide in the liver, which caused the toxicity issues (J. Appl. Toxicol., DOI: 10.1002/jat.1622).
Several other p38 blockers met a similar end. But while the p38 failures were “a major surprise and unexpected,” according to a 2009 review article (Ann. Rheum. Dis., DOI: 10.1136/ard.2009.119479), not all has been lost. Researchers are now looking at kinases upstream of p38 in inflammation signaling as potentially more effective drug targets. In 2011, a team in Europe used doramapimod as a starting point toward developing blockers of another kinase associated with lymphoma (ChemMedChem, DOI: 10.1002/cmdc.201100168).



Join the conversation
Contact the reporter
Submit a Letter to the Editor for publication
Engage with us on Twitter