Advertisement
Grab your lab coat. Let's get started
Welcome!
Welcome!
Create an account below to get 6 C&EN articles per month, receive newsletters and more - all free.
It seems this is your first time logging in online. Please enter the following information to continue.
As an ACS member you automatically get access to this site. All we need is few more details to create your reading experience.
Not you? Sign in with a different account.
Not you? Sign in with a different account.
ERROR 1
ERROR 1
ERROR 2
ERROR 2
ERROR 2
ERROR 2
ERROR 2
Password and Confirm password must match.
If you have an ACS member number, please enter it here so we can link this account to your membership. (optional)
ERROR 2
ACS values your privacy. By submitting your information, you are gaining access to C&EN and subscribing to our weekly newsletter. We use the information you provide to make your reading experience better, and we will never sell your data to third party members.
Chemical Bonding
Hydrogen Bonds Visualized
Microscopy: AFM reveals details of intermolecular interactions
by Jyllian Kemsley
September 30, 2013
| A version of this story appeared in
Volume 91, Issue 39
\
\
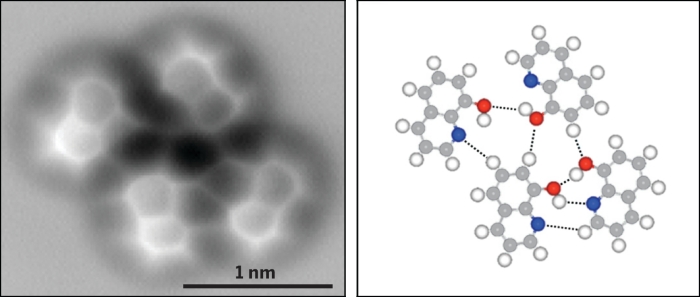
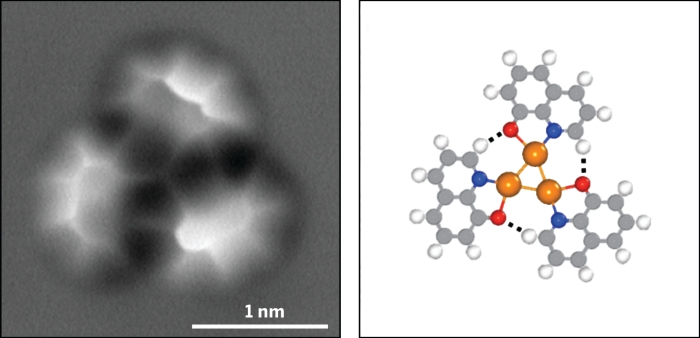
AFM images of 8-hydroxyquinoline on a copper surface show hydrogen-bonding interactions at low temperatures (top) and room temperature (bottom); C = gray, H = white, O = red, N = blue, Cu = orange.
Hydrogen bonds are ubiquitous—and universally important—in chemistry. They give water its unique properties, speed or slow reactions, and hold together the three-dimensional shapes of DNA, proteins, and other supramolecular structures. But chemists have never actually seen them.
Now, with high-resolution atomic force microscopy (AFM), they have (Science 2013, DOI: 10.1126/science.1242603). The stunning images should enhance understanding of hydrogen bonds’ hitherto elusive properties.
AFM has produced several pinup molecular images over the past several years—showing the atoms and bonds in single molecules of pentacene, revealing the bond order of complex molecules, and exposing before and after pictures of molecules undergoing a chemical reaction. Hydrogen bonds are another big win for the technique.
Roughly defined, these bonds are weak attractive interactions between an atom or group of atoms and a hydrogen that is covalently bonded to an atom that is more electronegative than hydrogen (Pure Appl. Chem. 2011, DOI: 10.1351/pac-rec-10-01-02). Hydrogen bonds form when lone pairs of electrons or even those within π clouds are attracted to the slightly electropositive hydrogens, although not strongly enough to form conventional covalent bonds.
In the new work, a team led by chemistry and physics professors Xiaohui Qiu and Zhihai Cheng of China’s National Center for Nanoscience & Technology and physics professor Wei Ji of Renmin University of China used a technique called noncontact AFM to look for intermolecular forces between a variety of compounds. They succeeded with 8-hydroxyquinoline deposited on a copper surface.
At temperatures near absolute zero, the researchers found that 8-hydroxyquinoline formed hydrogen-bonded aggregates, with the electron density of the hydrogen bonds made visible by AFM. Seeing the hydrogen bonds was unexpected, Qiu says, because they have very low electron density. “It was definitely a very exciting moment,” he says.
At room temperature, the researchers also observed hydrogen bonds in dimers and trimers of 8-hydroxyquinoline radicals complexed with copper. The species likely formed through a previously known dehydrogenation reaction of hydroxyl groups on copper surfaces.
“I think this work is seminal,” says Leo Gross, a staff researcher at IBM’s Zurich research center. Gross leads a team that visualized pentacene and has carried out several AFM studies since. He notes that combining interaction forces measured by AFM with density functional theory could yield better understanding of the properties and mechanism of hydrogen bonds.
Some scientists contend hydrogen bonds contain some covalent character, and AFM studies could provide new insights in that debate, says Elangannan Arunan, a chemistry professor at the Indian Institute of Science. Arunan led the International Union of Pure & Applied Chemistry committee that proposed a new definition of the hydrogen bond in 2010.


Join the conversation
Contact the reporter
Submit a Letter to the Editor for publication
Engage with us on Twitter