Advertisement
Grab your lab coat. Let's get started
Welcome!
Welcome!
Create an account below to get 6 C&EN articles per month, receive newsletters and more - all free.
It seems this is your first time logging in online. Please enter the following information to continue.
As an ACS member you automatically get access to this site. All we need is few more details to create your reading experience.
Not you? Sign in with a different account.
Not you? Sign in with a different account.
ERROR 1
ERROR 1
ERROR 2
ERROR 2
ERROR 2
ERROR 2
ERROR 2
Password and Confirm password must match.
If you have an ACS member number, please enter it here so we can link this account to your membership. (optional)
ERROR 2
ACS values your privacy. By submitting your information, you are gaining access to C&EN and subscribing to our weekly newsletter. We use the information you provide to make your reading experience better, and we will never sell your data to third party members.
Pharmaceuticals
Taking Multiple Shots At Drug-Resistant Cancer
Medicinal Chemistry: Technique directly links fragments to make a variety of small molecules that block hot target Mcl-1
by Carmen Drahl
January 8, 2013

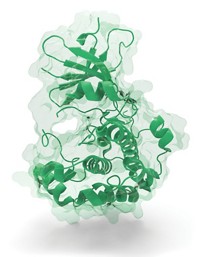
Several types of cancers, including many of those resistant to chemotherapy, boost levels of a cell-survival protein called myeloid cell leukemia 1 (Mcl-1). Though shutting down Mcl-1 has become a popular quest for medicinal chemists, none of the blocking molecules they’ve developed has reached human clinical trials. Now, researchers report a strategy that provides new Mcl-1 blockers (J. Med. Chem., DOI: 10.1021/jm301448p). The molecules themselves are only starting points for drugs, the researchers say, but the technique carries power in its ability to provide multiple shots on goal—a variety of candidates for anticancer drugs.
Vanderbilt University School of Medicine’s Stephen W. Fesik has known about Mcl-1 and the difficulties targeting it since the 1990s when he worked on cancer drug discovery at Abbott Laboratories. He and his Vanderbilt colleagues decided to apply their specialty, a technique that uses NMR to detect binding interactions between proteins and small molecules, to the problem. They searched a library of chemical fragments which typically bind weakly to biological targets, and found two families of molecules that bound to Mcl-1in neighboring spots on the protein.
Medicinal chemists working with fragments link them together to form better binders. However, NMR and molecular modeling suggested a slightly different strategy to Fesik’s team. Instead of using a linker to connect the two fragment classes, they designed a set of molecules with the fragments merged together. The researchers could do so because each fragment had a carboxylic acid that performed the same job--interacting with a single amino acid in Mcl-1.
Merging produces molecules that are great binders and that are small in size, Fesik explains. Because there tends to be an upper limit on how big drug molecules can be, the merged molecules’ size leaves more room for additions Importantly, Fesik adds, the compounds bind Mcl-1 selectively over related proteins.
By binding to Mcl-1, Fesik’s compounds prevent it from interacting with another protein in cancer cells. Such protein-protein interactions are typically tough targets for medicinal chemists because the interactions often involve shallow protein pockets that don’t lend themselves to tight binding with small molecules, explains Maurizio Pellecchia of Sanford-Burnham Medical Research Institute, who wrote a commentary on the work (DOI: 10.1021/jm301837n). Fesik’s NMR method, however, finds strong binders that burrow deep into pockets by inducing small changes in Mcl-1’s conformation, thus preventing the protein-protein interactions.
“This work is solid from top to bottom,” adds fragment-based drug discovery expert James Wells of the University of California, San Francisco.
Even though Fesik is refining his series of compounds for testing in mice, he cautions that drug discovery is rife with failures. Still, the screen turned up nine other sets of fragments, he says, “so if these should fail we have other options for designing clinically useful Mcl-1 inhibitors. This is just the tip of the iceberg.”


Join the conversation
Contact the reporter
Submit a Letter to the Editor for publication
Engage with us on Twitter